S Orbital Drawing
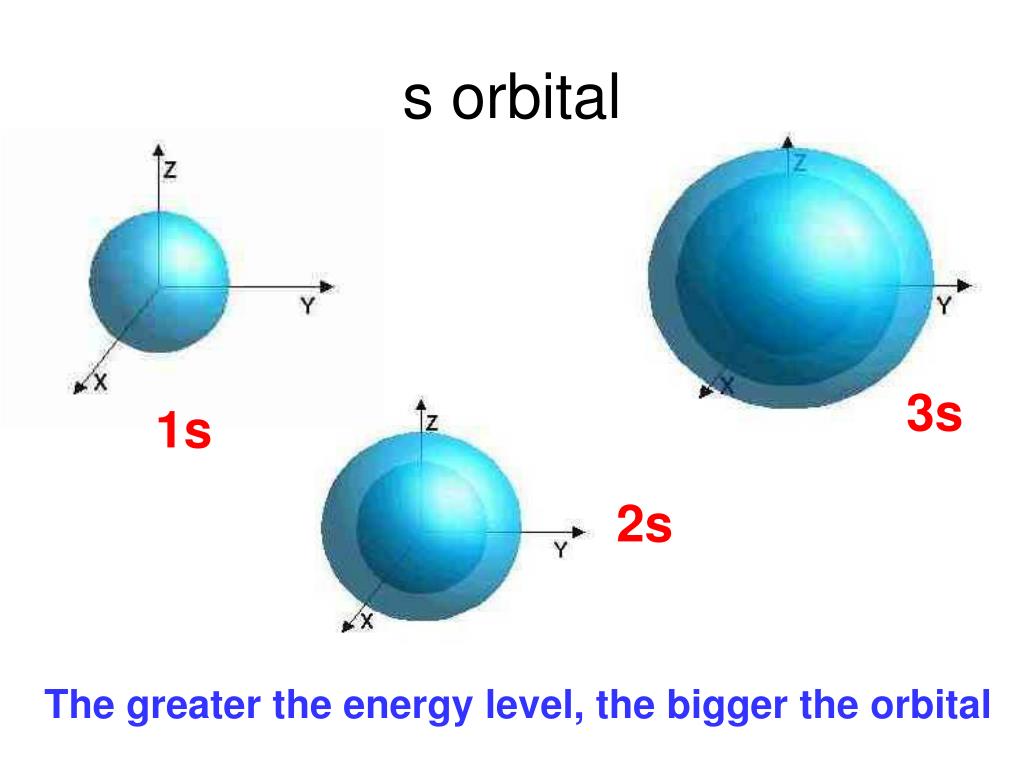
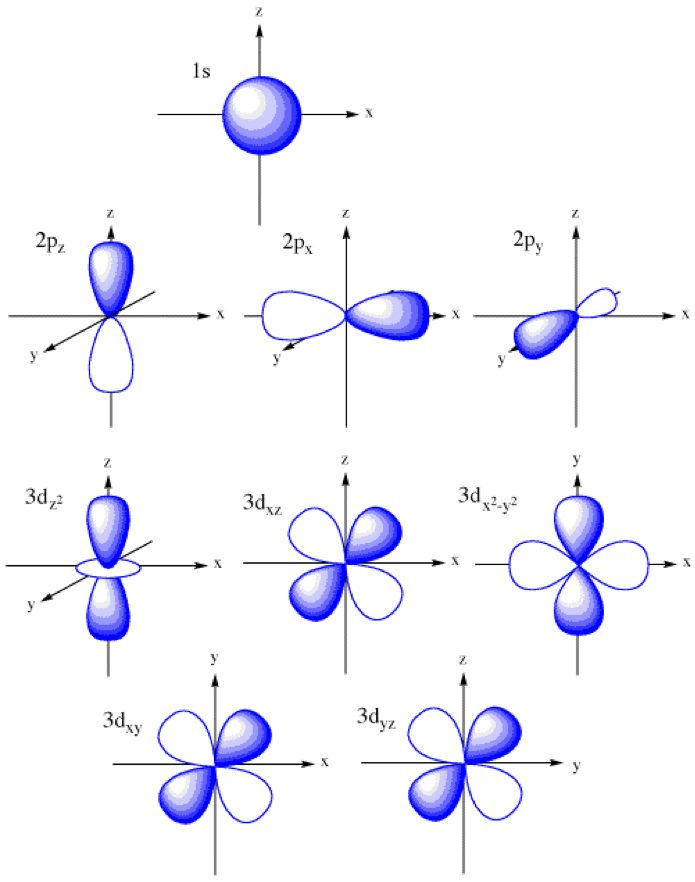
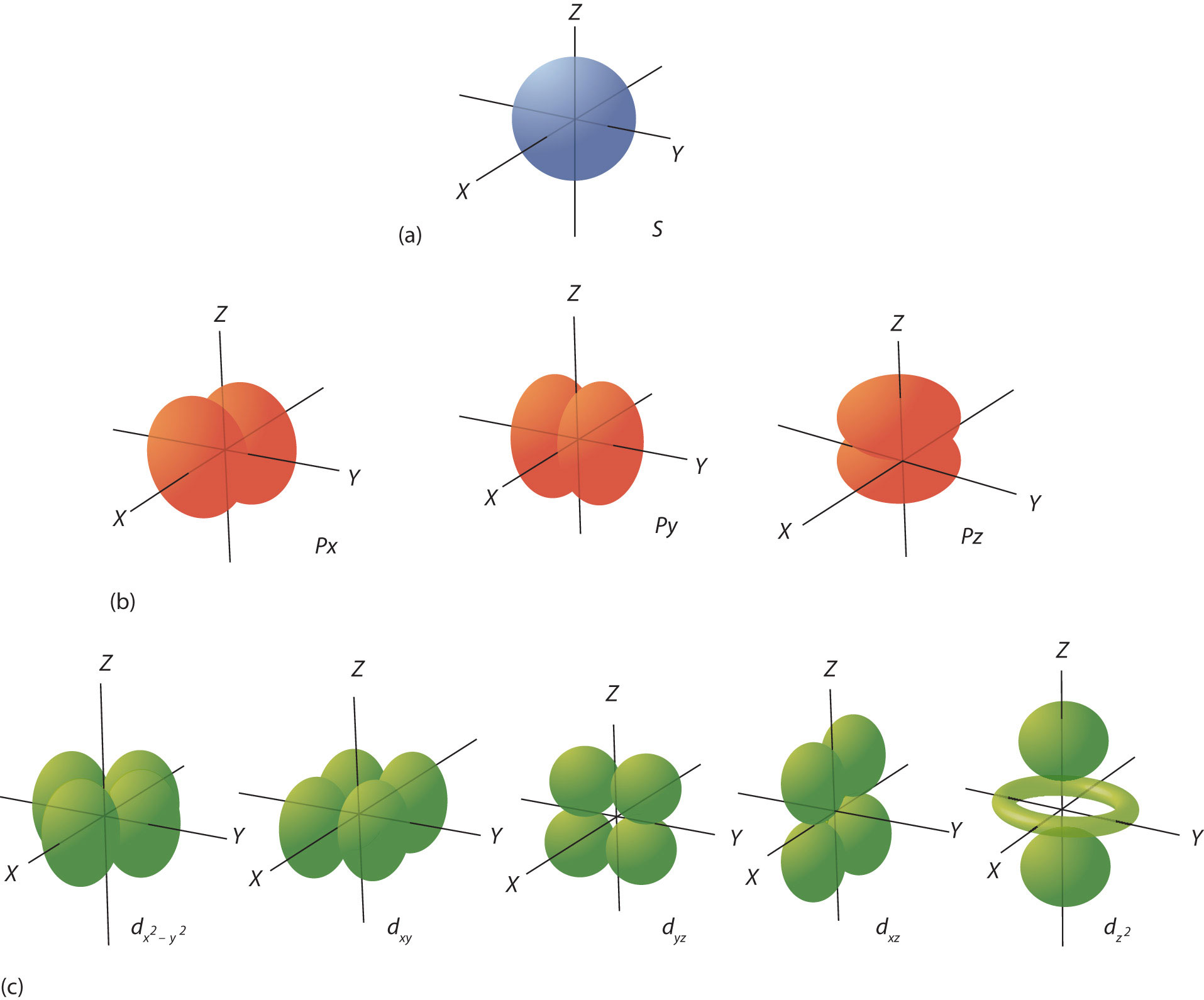
S Orbital Drawing - In a typical drawing of orbital, we first plot the radial wave function and the angular part is superimposed. Notice that the s orbital always has a slightly lower energy than the p orbitals at the same energy level, so the s orbital always fills with electrons before the corresponding p orbitals. This function describes the electron's charge distribution around the atom's nucleus, and can be used to calculate the probability of finding an electron in a specific region around the nucleus. Therefore, s orbital only has radial nodes, which are spheres. Web all we can do is draw a shape that will include the electron most of the time, say 95% of the time. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. For the p orbitals, draw one arrow pointing up on each of the lines first. The lower grades have around 33 students on average. This process is the same for the d and f orbitals. Web orbitals and orbits. Web the boundary surface diagram for the s orbital looks like a sphere having the nucleus as its centre which in two dimensions can be seen as a circle. The shape of the orbital depends on the quantum numbers associated with an energy state. Web all we can do is draw a shape that will include the electron most of. The shapes of some typical orbitals are discussed below. In a typical drawing of orbital, we first plot the radial wave function and the angular part is superimposed. This means that the first shell can hold 2 electrons. All s orbitals have l = m = 0, but the value of n can vary. Then, fill the lines with an. This means that the first shell can hold 2 electrons. Web types of atomic orbitals. This process is the same for the d and f orbitals. We classified the different orbital into shells and sub shells to distinguish them more easily. Zygomaticofadalis 8j landmarks and measurements on anterior aspect of the head and neck (1) the surface landmarks related to. We call this shape the 95% contour. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus. Upper grade text books do not have the additional picture cards and require more imagination to teach the unit. The shape of the s orbital is a sphere; A. Figure eight shaped orbital with two lobes and one node. Notice that the s orbital always has a slightly lower energy than the p orbitals at the same energy level, so the s orbital always fills with electrons before the corresponding p orbitals. Spherical shaped orbital with no nodes. When a planet moves around the sun, its definite path, called. The shape of the orbital depends on the quantum numbers associated with an energy state. This is also due to the history when they were discovered. The angular nodes is l, which is 0 for all s orbitals; Notice that the s orbital always has a slightly lower energy than the p orbitals at the same energy level, so the. For a p orbital, draw a figure eight; Imagine shells around the nucleus, that get bigger and bigger. The real oddity is the position of the 3d orbitals. For an s orbital, draw a circle; Web depict the orbital shapes are intended to describe the region encompassing 90−95% probability density. Get a 10 bullets summary of the topic. An orbital is a space where a specific pair of electrons can be found. Web the boundary surface diagram for the s orbital looks like a sphere having the nucleus as its centre which in two dimensions can be seen as a circle. A p orbital consists of two lobes of electron. A drastically simplified view of the atom looks similar, in which the electrons orbit around the nucleus. Web founded in 2010, with the mission of continuously improving the intelligence, precision, safety and convenience of the laboratory, infitek is a professional manufacturer driven by innovation and service in laboratory and medical field, our company is certified by iso9001, iso13485 and intellectual. For a p orbital, draw a figure eight; Therefore, s orbital only has radial nodes, which are spheres. In a typical drawing of orbital, we first plot the radial wave function and the angular part is superimposed. Web all we can do is draw a shape that will include the electron most of the time, say 95% of the time.. Get a 10 bullets summary of the topic. Web depict the orbital shapes are intended to describe the region encompassing 90−95% probability density. Web the boundary surface diagram for the s orbital looks like a sphere having the nucleus as its centre which in two dimensions can be seen as a circle. They are at a slightly higher level than the 4s. Every orbital is a shape (that can be determined by a trigonometric function i. A p orbital consists of two lobes of electron density on either side of the nucleus. The angular nodes is l, which is 0 for all s orbitals; An s orbital is spherically symmetric around the nucleus of the atom, like a hollow ball made of rather fluffy material with the nucleus at its centre. The shape of the s orbital is a sphere; In two dimensions, we draw it as a circle. There are three common orbital types: Web founded in 2010, with the mission of continuously improving the intelligence, precision, safety and convenience of the laboratory, infitek is a professional manufacturer driven by innovation and service in laboratory and medical field, our company is certified by iso9001, iso13485 and intellectual property management system. The real oddity is the position of the 3d orbitals. This is also due to the history when they were discovered. Electrons, in fact, inhabit regions of space known as orbitals. Web for a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus.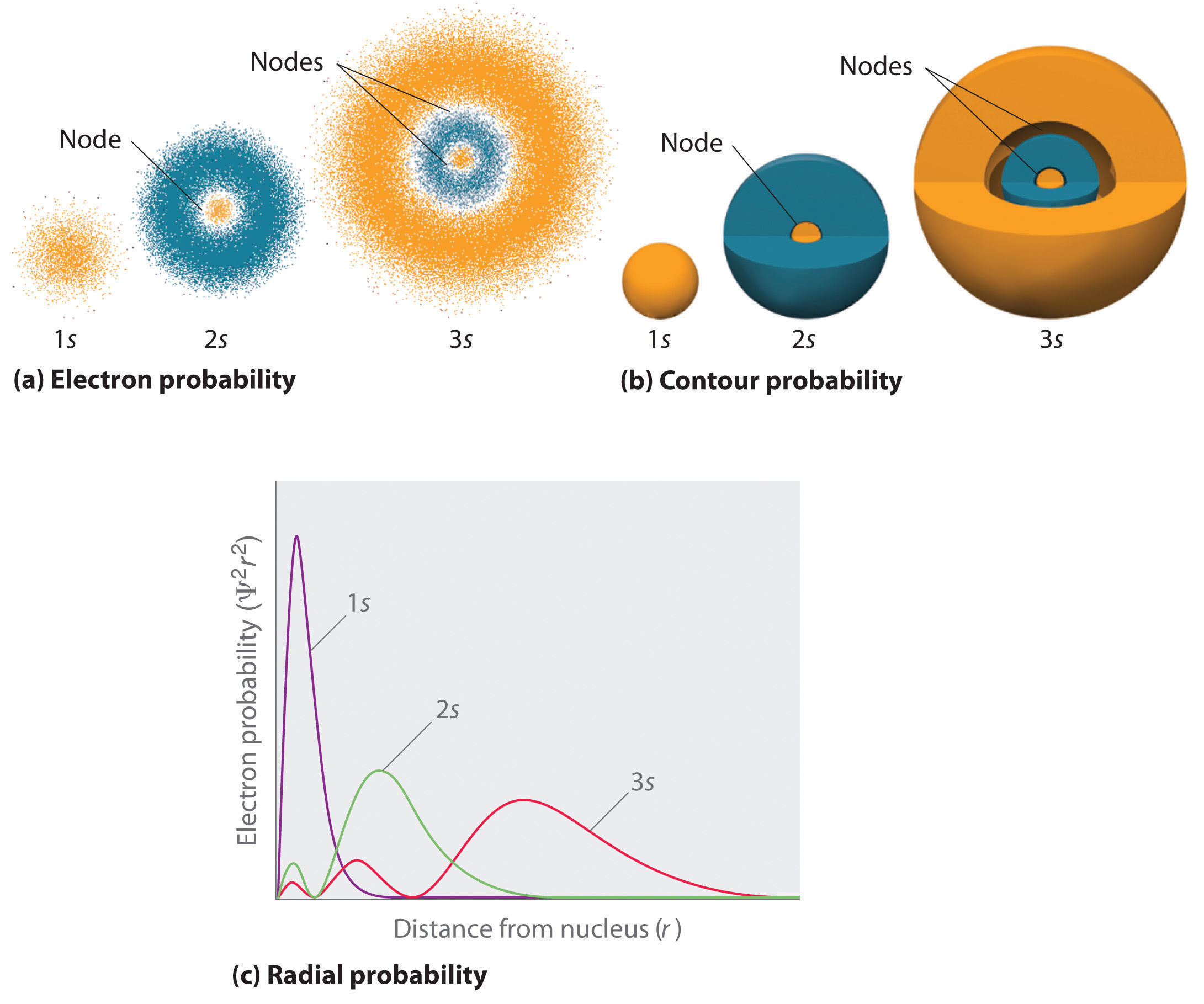
s Atomic Orbitals Chemistry LibreTexts
PPT Chapter 5 Electrons In Atoms PowerPoint Presentation, free
Atomic orbitals explained polizhuge
3.7 Electron Arrangement The Quantum Model Chemistry LibreTexts

Shapes of Atomic Orbitals — Overview & Examples Expii
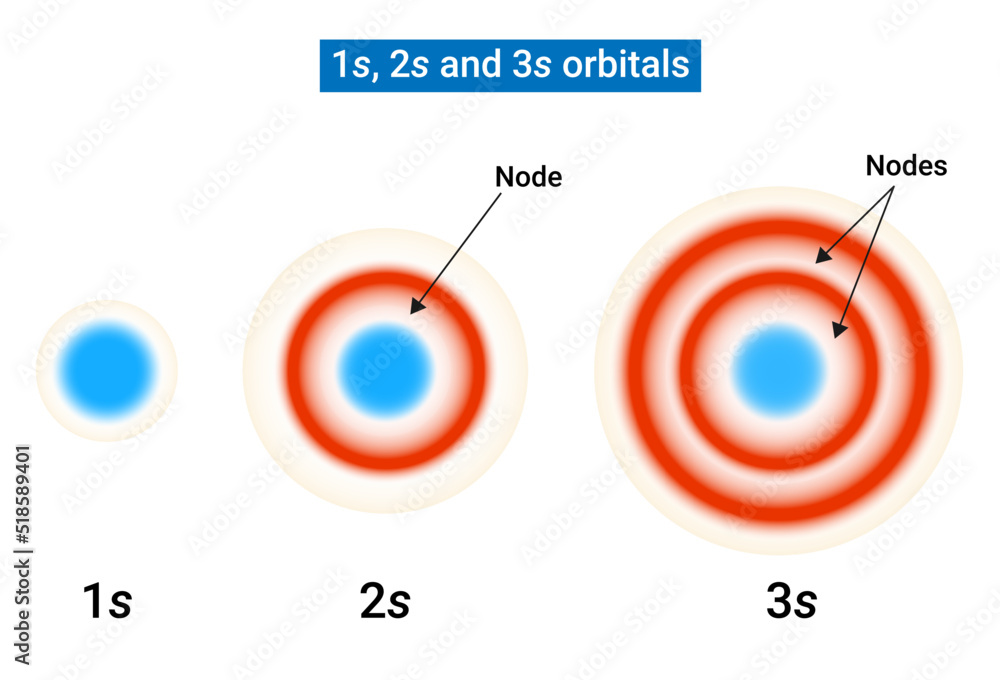
Shapes of Atomic Orbital s orbitals radial nodes 1s, 2s and 3s Stock
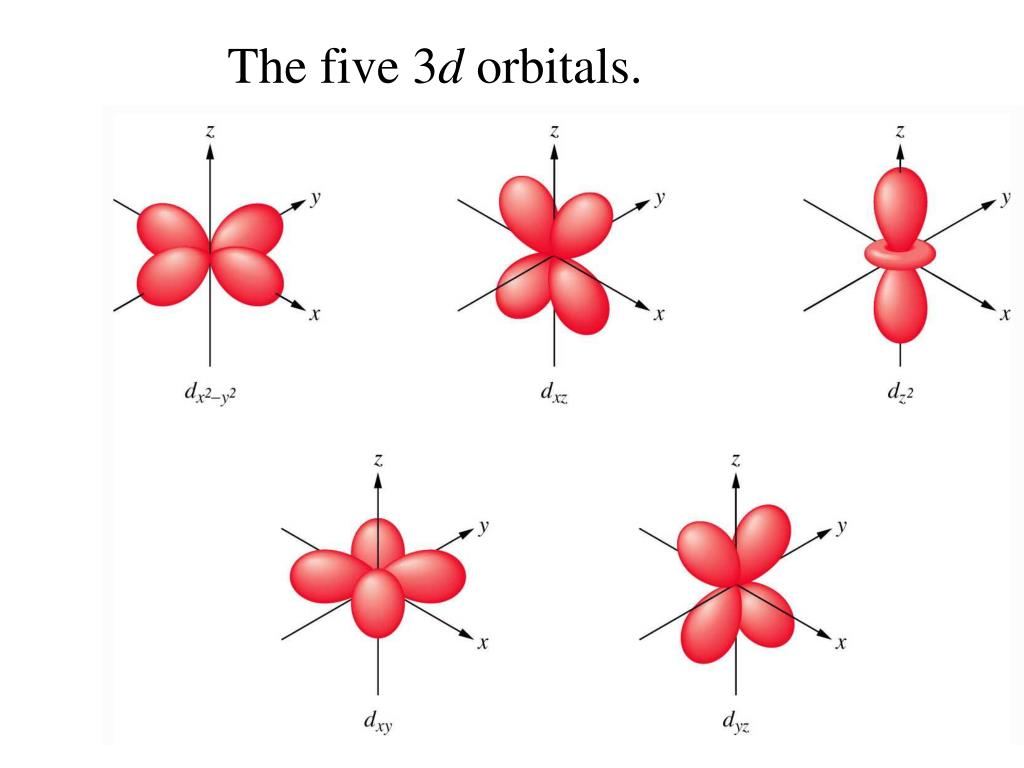
PPT Cutaway diagrams showing the spherical shape of S orbitals

Classify these atomic orbitals innovativehrom

Biochemistry Glossary Orbitals 2. Shape Draw It to Know It

12.1.5 Draw the shape of an s orbital and the shapes of the p x , p y
This Means That The First Shell Can Hold 2 Electrons.
Then, Fill The Lines With An Arrow Pointing Down, Until The Number Of Arrows Drawn Is Equal To The Electron Occupancy.
S Orbitals Are Spherically Symmetric.
This Function Describes The Electron's Charge Distribution Around The Atom's Nucleus, And Can Be Used To Calculate The Probability Of Finding An Electron In A Specific Region Around The Nucleus.
Related Post: