P Orbital Drawing
P Orbital Drawing - Because the 2 p subshell has l = 1, with three values of ml (−1, 0, and +1), there are three 2 p orbitals. This is called a p (pi) bond. As the value of l increases, the number of orbitals in a given subshell increases, and the shapes of the orbitals become more complex. Web for the p orbitals, draw one arrow pointing up on each of the lines first. Each p orbital consists of two sections better known as lobes which lie on either side of the plane passing through the nucleus. Label the positions of the oxygen nuclei with the symbol o. Web the 1 s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. As the value of l increases, the number of orbitals in a given subshell increases, and the shapes of the orbitals become more complex. The three p orbitals are at right angles to each other and have a lobed shape. If there are more electrons after the 1s, and 2s orbitals have been filled, each p orbital will be filled with one electron first before two electrons try to reside in the same p orbital. Web subshells are designated by the letters s , p , d , and f , and each letter indicates a different shape. Then, fill the lines with an arrow pointing down, until the number of arrows drawn is equal to the electron occupancy. It only has s s orbitals. As the value of l increases,. There is a zero probability of finding the electron on that plane. The energy increases as we move up to the 2 s and then 2 p, 3 s, and 3 p orbitals, showing that the increasing n value has more influence on energy than the increasing l value for small atoms. The resulting orbitals contain nodes along the bond. Click here:point_up_2:to get an answer to your question :writing_hand:draw the structure of porbitals. Web for example, the 2p shell has three p orbitals. 400 views 9 months ago electron configuration. The size of the p orbitals also increases as the energy level or shell increases. Each p orbital consists of two sections better known as lobes which lie on either. An orbital is a space where a specific pair of electrons can be found. Web 68.draw the structure of molecular orbital of o2. As such, the first shell has no p p orbitals; The three p orbitals are at right angles to each other and have a lobed shape. Web p orbitals (l=1) only s orbitals are spherically symmetrical. The three p orbitals are at right angles to each other and have a lobed shape. This process is the same for the d and f orbitals. Web subshells are designated by the letters s , p , d , and f , and each letter indicates a different shape. The three p orbitals differ in. It only has s s orbitals. Web 68.draw the structure of molecular orbital of o2. The orbital shows where there is a 95% chance of finding a particular electron. As the value of l increases, the number of orbitals in a given subshell increases, and the shapes of the orbitals become more complex. We classified the different orbital into shells. Imagine a horizontal plane through the nucleus, with one lobe of the orbital above the plane and the other beneath it; The orbital shows where there is a 95% chance of finding a particular electron. Web subshells are designated by the letters s , p , d , and f , and each letter indicates a. Want to join the conversation? Remember that l l must always be less than n n. Phosphorus has the symbol p and “it has a concentration in the earth’s crust of about one gram per kilogram”. Click here:point_up_2:to get an answer to your question :writing_hand:draw the structure of porbitals. Different lobes of p orbitals are often drawn for convenience as. As the value of l increases, the number of orbitals in a given subshell increases, and the shapes of the orbitals become more complex. Want to join the conversation? Because the 2 p subshell has l = 1, with three values of ml (−1, 0, and +1), there are three 2 p orbitals. In this video, we will see what. In two dimensions, we draw it as a circle. However, this pattern does not hold for larger atoms. A p orbital along the y axis is labeled p y and one along the z axis is a p z orbital. The size of the p orbitals also increases as the energy level or shell increases. Web for example, the 2p. An orbital is a space where a specific pair of electrons can be found. For an f orbital, see below. There is a zero probability of finding the electron on that plane. The three p orbitals differ in the way the lobes are oriented whereas they are identical in. A p orbital along the y axis is labeled p y and one along the z axis is a p z orbital. The illustration above is for one set of p orbitals that are orthogonal to the bond axis. Orbitals with total angular momentum quantum number l = 1 l = 1 are called p p orbitals. If there are more electrons after the 1s, and 2s orbitals have been filled, each p orbital will be filled with one electron first before two electrons try to reside in the same p orbital. In the video.why does 1 electron from the s orbital and 2 electrons from the p orbitals combine? 400 views 9 months ago electron configuration. In sp² hybridization, one s orbital and two p orbitals hybridize to form three sp² orbitals, each consisting of 33% s character and 67% p character. Then, fill the lines with an arrow pointing down, until the number of arrows drawn is equal to the electron occupancy. Web the shape of p orbitals. As such, the first shell has no p p orbitals; In this video, we will see what orbital diagrams are, the rules that we follow while determining one for any element and. This is called a p (pi) bond.
Electron Orbitals
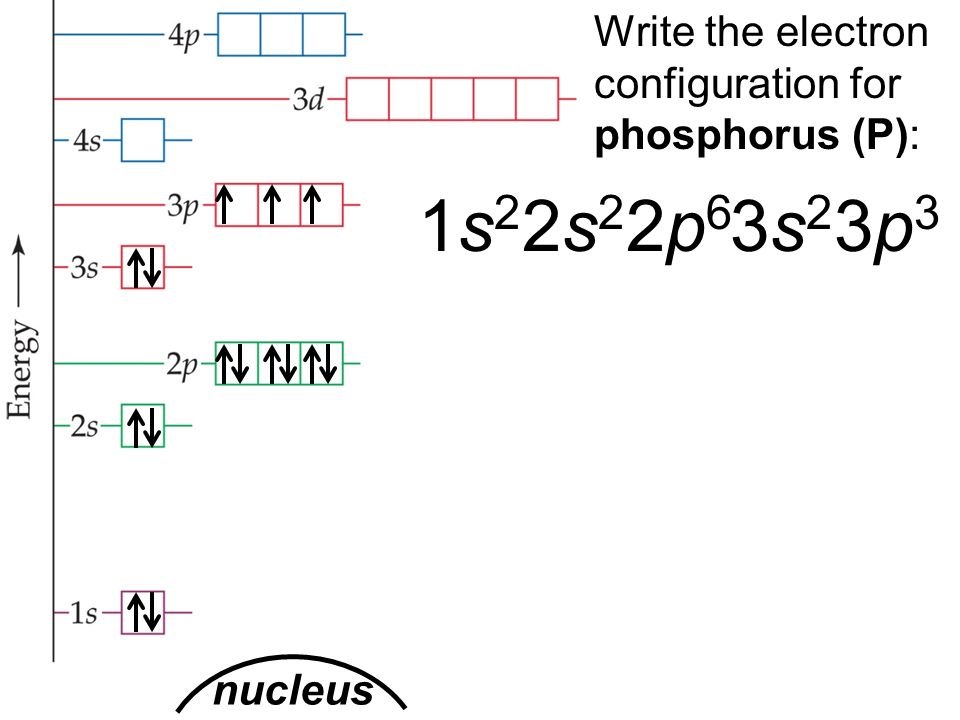
Phosphorus Electron Configuration (P) with Orbital Diagram
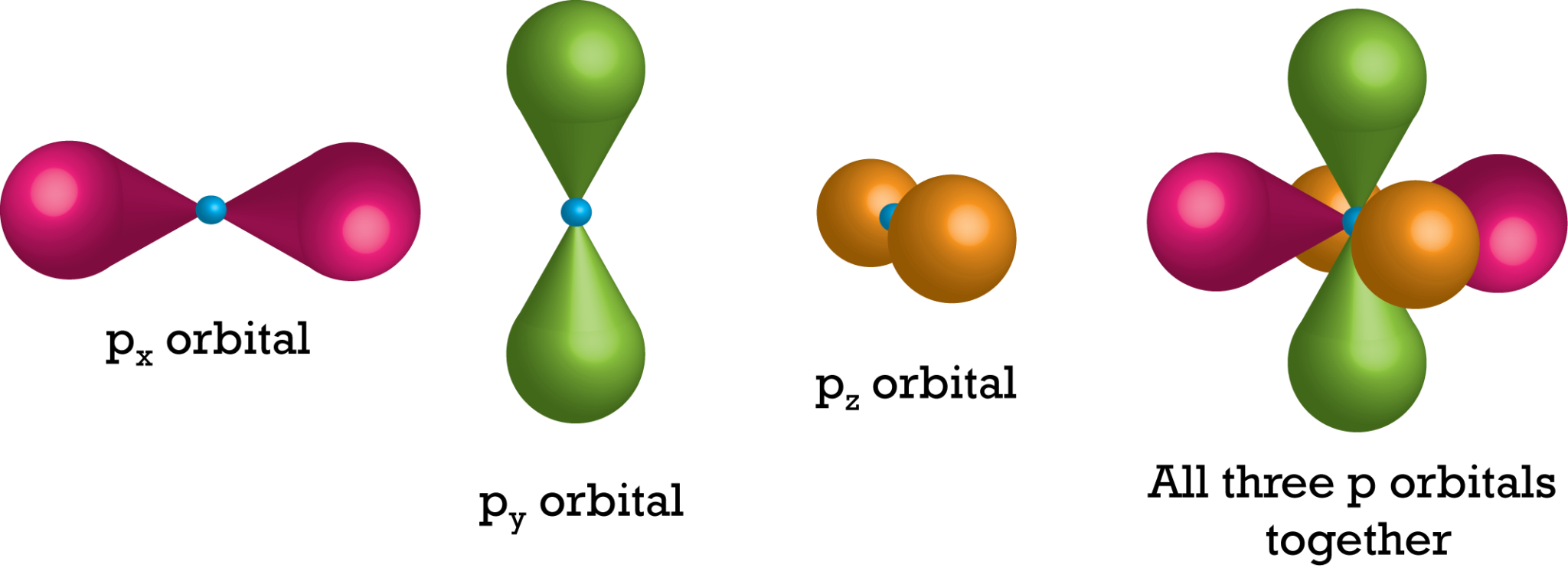
Shapes of Orbitals and their Types Chemistry Skills
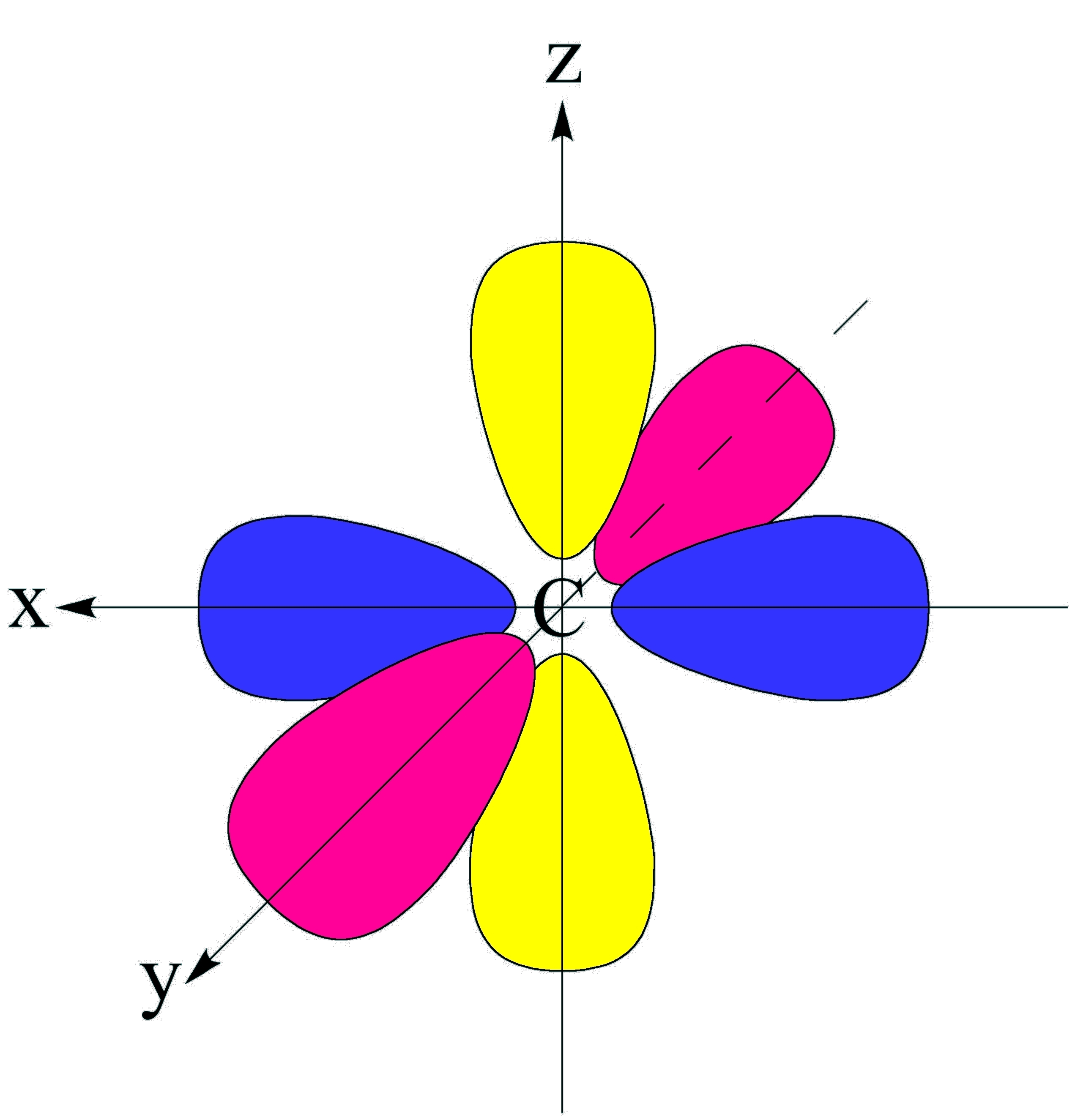
Illustrated Glossary of Organic Chemistry Orbital

Molecular Orbitals Introductory Chemistry
[Solved] sketch sigma and pi bond from p orbital Course Hero

12.1.5 Draw the shape of an s orbital and the shapes of the p x , p y

P orbital The Student Room

Shapes of Atomic Orbitals — Overview & Examples Expii
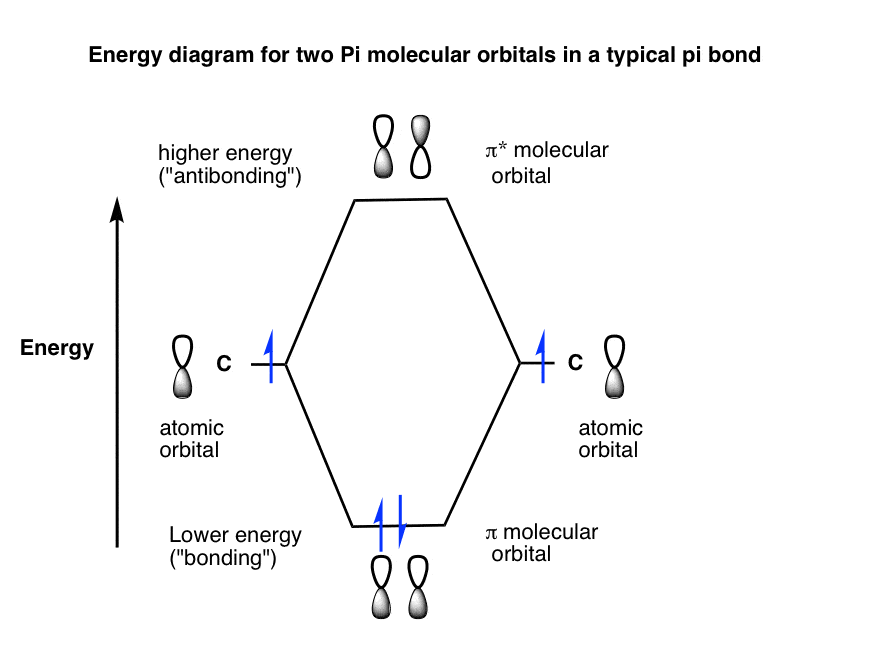
Bonding And Antibonding Pi Orbitals Master Organic Chemistry
An S Orbital Is A Sphere.
This Is Also Due To The History When They Were Discovered.
Web P Orbitals (L=1) Only S Orbitals Are Spherically Symmetrical.
A P Orbital Consists Of Two Lobes Of Electron Density On Either Side Of The Nucleus.
Related Post: