How To Draw An Ion
How To Draw An Ion - Web the organic chemistry tutor. The diagram is also called a lewis dot diagram, lewis. 152k views 3 years ago. We need to draw another resonance structure. Only give reasonable results for covalent compounds and polyatomic ions of the main group (s and p block) elements, can not predict the structure of ionic compounds, Web lewis structures of ions. Web this tutorial shows you how to convert diagrams of atoms into diagrams of ions. (1*5) + (3*6) + 1(ion) = 24 electrons. When drawing the structure of an ion, be sure to add/subtract electrons to account for. Many of the ions that form have eight electrons in their valence shell. Count up the valence electrons: When discussing the octet rule, we do not consider d or f. Practice with drawing lewis structures. Web hence it is very important to learn how to draw lewis dot structure correctly for an atom, ion, molecule, polyatomic ion and an ionic compound. Shared pairs of electrons are drawn as lines between atoms, while lone. Not even under a complex microscopic can we view the individual electrons that surround an atom’s nuclei. The three oxygens are drawn in the shape of a triangle with the nitrogen at the center of the triangle. Magnesium is a metal in group two of the periodic table, so will form a 2 + ion. 152k views 3 years ago.. We need to draw another resonance structure. Web the example is for the nitrate ion. Web lewis structures of ions. The diagram is also called a lewis dot diagram, lewis. Web draw lewis structures for ionic compounds. Draw a lewis electron dot diagram for an atom or a monatomic ion. 292k views 3 years ago new ap & general chemistry video playlist. When atoms have fewer than eight electrons, they tend to react and form more stable compounds. This chemistry video explains how to draw the lewis structures of ionic compounds. In section 4.7, we demonstrated that. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. We need to draw another resonance structure. Find the element on the periodic table and identify the group number at the top of the column. Now that you’ve mastered the art of drawing the lewis structures of neutral covalent compounds, it’s time to draw the. Many of the ions that form have eight electrons in their valence shell. Not even under a complex microscopic can we view the individual electrons that surround an atom’s nuclei. We need to draw another resonance structure. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Find the element. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Magnesium is a metal in group two of the periodic table, so will form a 2 + ion. This video explains how to draw the electron configuration of an ion of sodium, as well as many other representations of. Add. 292k views 3 years ago new ap & general chemistry video playlist. Figure 1 a chloride ion note the square brackets to show what is charged and the numerical charge on the top right of the square brackets. Find the element on the periodic table and identify the group number at the top of the column. Draw the outer shell. (1*5) + (3*6) + 1(ion) = 24 electrons. Web the organic chemistry tutor. 152k views 3 years ago. For the no+ structure use the periodic table to find the total number of. Draw the outer shell of each atom. Web draw lewis structures for ionic compounds. 215k views 6 years ago. Magnesium is a metal in group two of the periodic table, so will form a 2 + ion. 292k views 3 years ago new ap & general chemistry video playlist. Figure out how many electrons the molecule must have, based on the number of valence electrons in each. Web lewis structures of ions. The octet rule refers to the tendency of atoms to prefer to have eight electrons in the valence shell. Web draw lewis structures for ionic compounds. Web determine the electron configuration of ions. Web bohr diagrams of atoms and ions. Count up the valence electrons: 215k views 6 years ago. Web this tutorial shows you how to convert diagrams of atoms into diagrams of ions. Draw a lewis electron dot diagram for an atom or a monatomic ion. 292k views 3 years ago new ap & general chemistry video playlist. How to draw a dot and cross diagram for magnesium oxide. Magnesium is a metal in group two of the periodic table, so will form a 2 + ion. A rule stating that atoms lose, gain, or share electrons in order to have a full valence shell of 8 electrons. Remember that lewis dot structures. If you haven’t mastered the art of drawing lewis structures yet, you can check out the tutorial by clicking here. The astute reader may have noticed something:
C4 Drawing ions YouTube
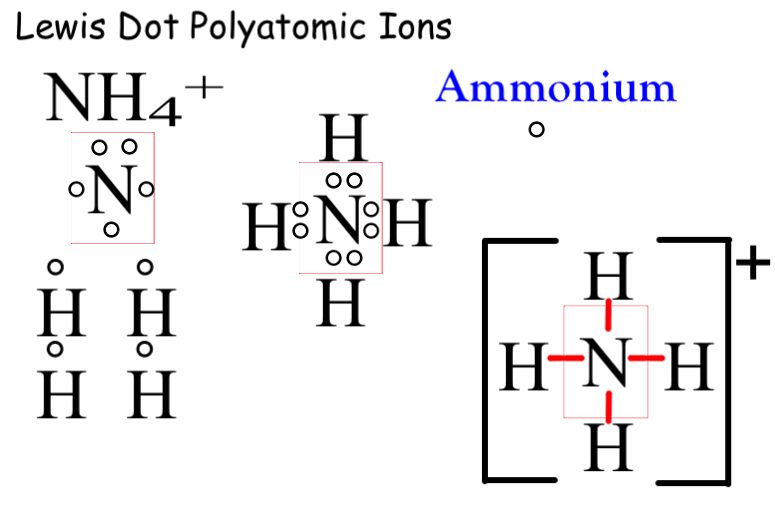
How do you draw lewis structures for polyatomic ions? Socratic

How To Draw Lewis Structures A Step By Step Tutorial

How to Draw an Ion YouTube
Ion atom molecule education poster Royalty Free Vector Image
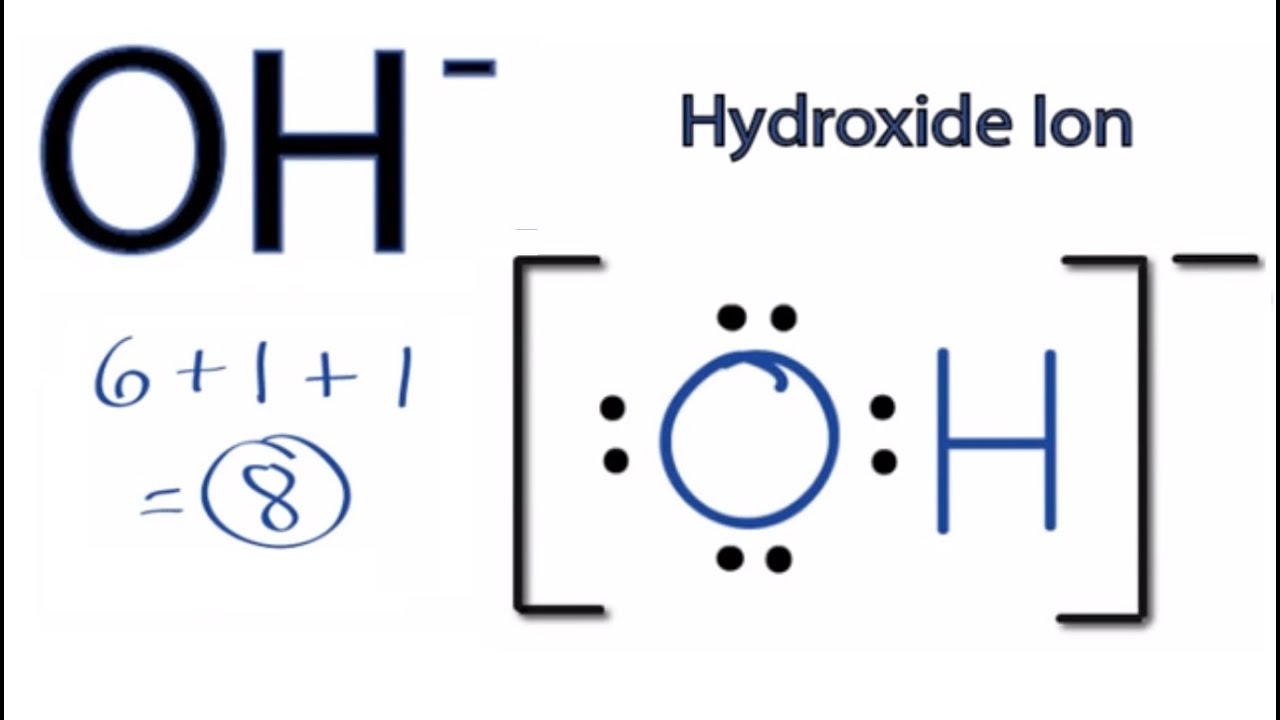
OH Lewis Structure How to Draw the Lewis Dot Structure for the

Review Drawing Atoms and Ions YouTube

How to draw ionic lewis dot structures YouTube

How to draw ion YouTube

Drawing Ions (NCEA L1 & Junior Science) YouTube
Not Even Under A Complex Microscopic Can We View The Individual Electrons That Surround An Atom’s Nuclei.
When Constructing A Lewis Diagram, Keep In Mind The Octet Rule, Which Refers To The Tendency Of Atoms.
When Atoms Have Fewer Than Eight Electrons, They Tend To React And Form More Stable Compounds.
When Discussing The Octet Rule, We Do Not Consider D Or F.
Related Post: