Drawing Ionic Bonds
Drawing Ionic Bonds - Swap the crosses for dots in one of your diagrams. When atoms have fewer than eight electrons, they tend to react and form more stable compounds. Opposite charges attract and like charges repel. Representing ionic solids using particulate models. The compound is often added to otherwise odorless fuels such as natural gas to help warn of gas leaks. Ionic bonding occurs when atoms lose and gain electrons to form ions and then the positively and negatively charged ions are attracted to each other. During ionic bonding the atoms form ions by magnesium has two electrons in its outer gaining or losing electrons to obtain a full outer shell, oxygen has six. Web draw dot and cross diagrams of ionic bonding and explain how an ionic lattice is held together. This chemistry video explains how to draw the lewis structures of ionic compounds. Many of the ions that form have eight electrons in their valence shell. 6.7k views 7 years ago edexcel. It is a type of chemical bond that generates two oppositely charged ions. Web draw dot and cross diagrams of ionic bonding and explain how an ionic lattice is held together. Shows how to draw lewis dot structures for ionic compounds. In section 4.7, we demonstrated that ions are formed by losing electrons to. During ionic bonding the atoms form ions by magnesium has two electrons in its outer gaining or losing electrons to obtain a full outer shell, oxygen has six. Web revision notes on 1.6.4 ionic bonds: Back in section 4.8, we discussed the formation of ions. Web draw the electron configuration diagram for each atom. Web 224k views 5 years ago. Back in section 4.8, we discussed the formation of ions. Web a structural drawing showing an ionic bond between a sodium cation, na + , and a chloride anion, cl − . A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Draw lewis structures for ionic compounds. For example, when two chlorine. Atoms interact with each other through the formation of chemical bonds. Representing ionic solids using particulate models. The astute reader may have noticed something: During ionic bonding the atoms form ions by magnesium has two electrons in its outer gaining or losing electrons to obtain a full outer shell, oxygen has six. Web draw dot and cross diagrams of ionic. Magnesium has two electrons in its outer shell, oxygen has six. These grades are the stepping stone to your future. Shows how to draw lewis dot structures for ionic compounds. Web define ionic bond. Ionic bonds result from the attraction between oppositely charged ions. The skeletal structure of ethanethiol is shown below. Opposite charges attract and like charges repel. Back in section 4.8, we discussed the formation of ions. Many of the ions that form have eight electrons in their valence shell. Draw the electron configuration diagram for each atom. Swap the crosses for shell. Shows how to draw lewis dot structures for ionic compounds. In section 4.7, we demonstrated that ions are formed by losing electrons to make cations, or by gaining electrons to form anions. Draw lewis structures for ionic compounds. State the limitations of a range of models used to represent ionic bonding. Web a structural drawing showing an ionic bond between a sodium cation, na + , and a chloride anion, cl − . Draw lewis structures for ionic compounds. Web we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions. The astute. When drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Representing ionic solids using particulate models. Web draw dot and cross diagrams of ionic bonding and explain how an ionic lattice is held together. Draw the electron configuration diagram for each atom. Ionic bonding is the. When atoms have fewer than eight electrons, they tend to react and form more stable compounds. Back in section 4.8, we discussed the formation of ions. Ionic bonding occurs when atoms lose and gain electrons to form ions and then the positively and negatively charged ions are attracted to each other. Web a structural drawing showing an ionic bond between. Many of the ions that form have eight electrons in their valence shell. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Swap the crosses for shell. State the limitations of a range of models used to represent ionic bonding. Ionic bonding is the complete transfer of valence electron (s) between atoms. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Magnesium has two electrons in its outer shell, oxygen has six. In section 4.7, we demonstrated that ions are formed by losing electrons to make cations, or by gaining electrons to form anions. Ionic bonds result from the attraction between oppositely charged ions. Web we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions. When drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. It is a type of chemical bond that generates two oppositely charged ions. Web revision notes on 1.6.4 ionic bonds: During ionic bonding the atoms form ions by magnesium has two electrons in its outer gaining or losing electrons to obtain a full outer shell, oxygen has six. Swap the crosses for dots in one of your diagrams. During ionic bonding the atoms form ions by gaining or losing electrons to obtain a full outer shell.
ionic bond Definition, Properties, Examples, & Facts Britannica

Ionic Bonds Dot & Cross Diagrams (1.6 4) Edexcel IGCSE Chemistry
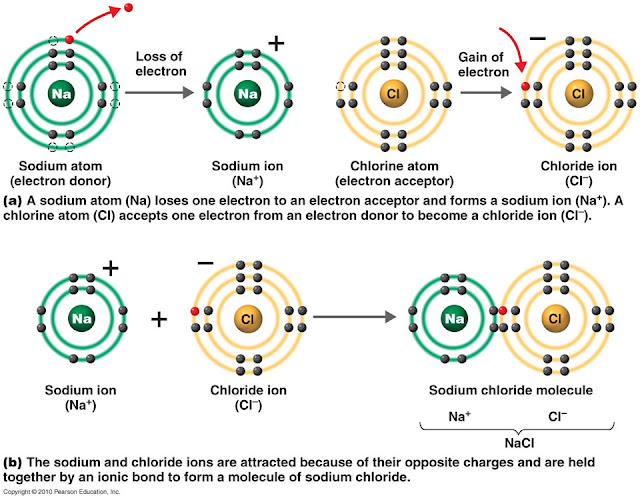
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Ionic Bonding Presentation Chemistry
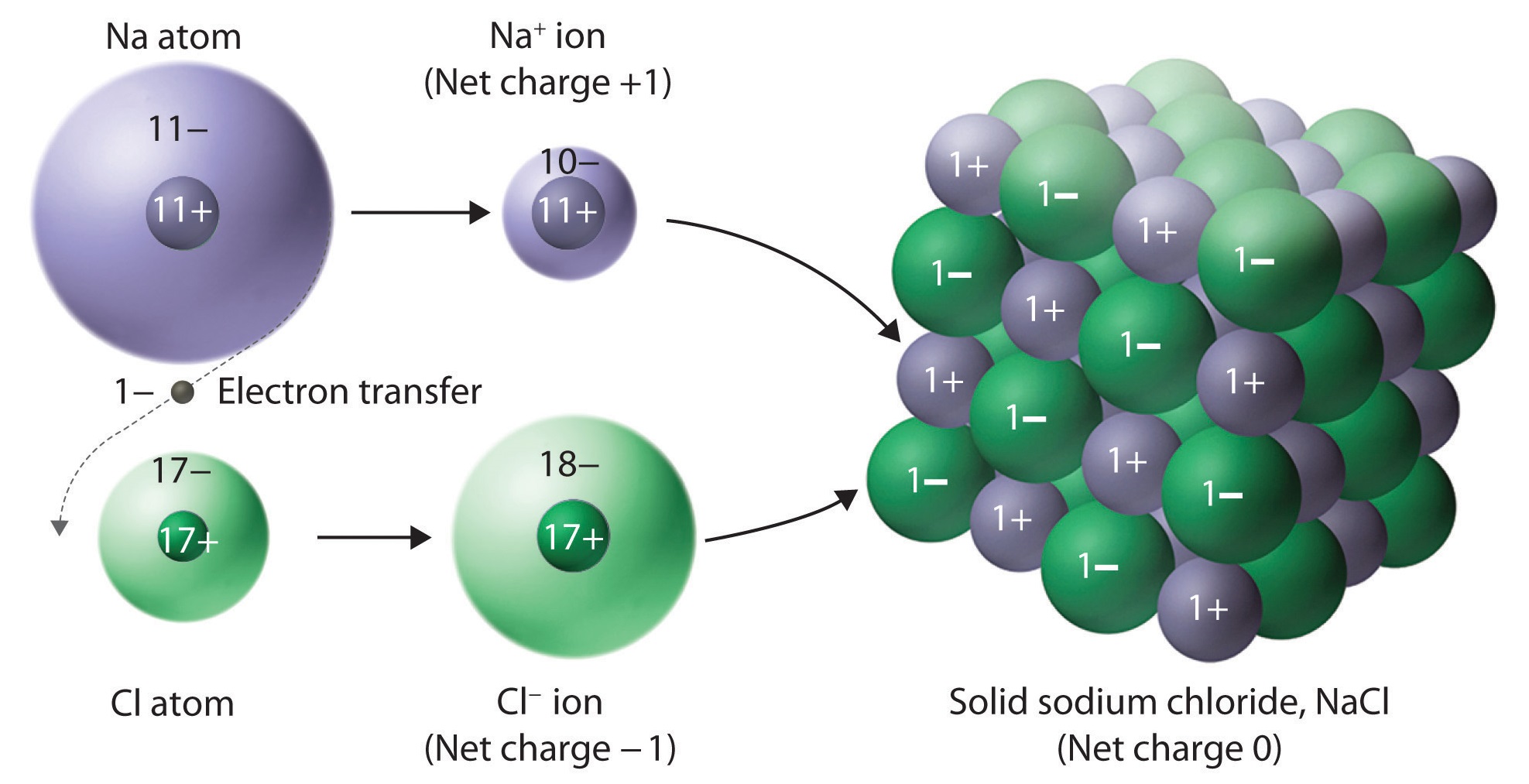
Ionic Solids Chemistry LibreTexts
:max_bytes(150000):strip_icc()/ionic-bond-58fd4ea73df78ca1590682ad.jpg)
Examples of Ionic Bonds and Compounds
Examples of Ionic Bonds and Compounds
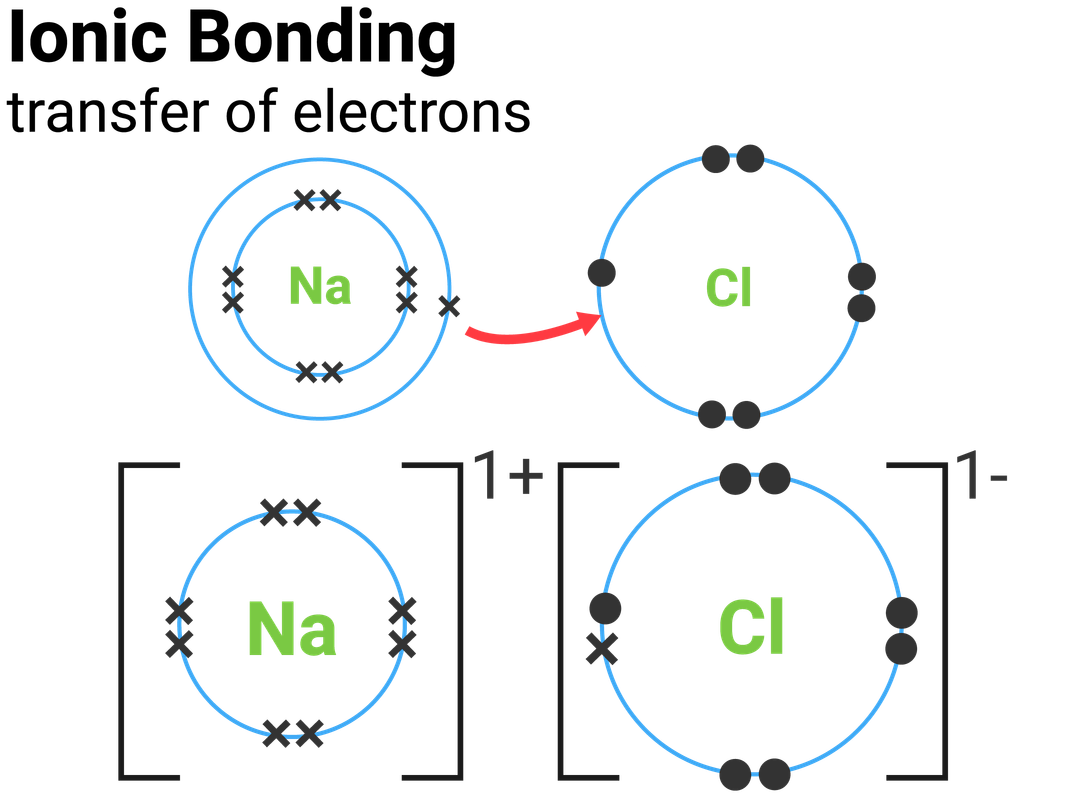
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk
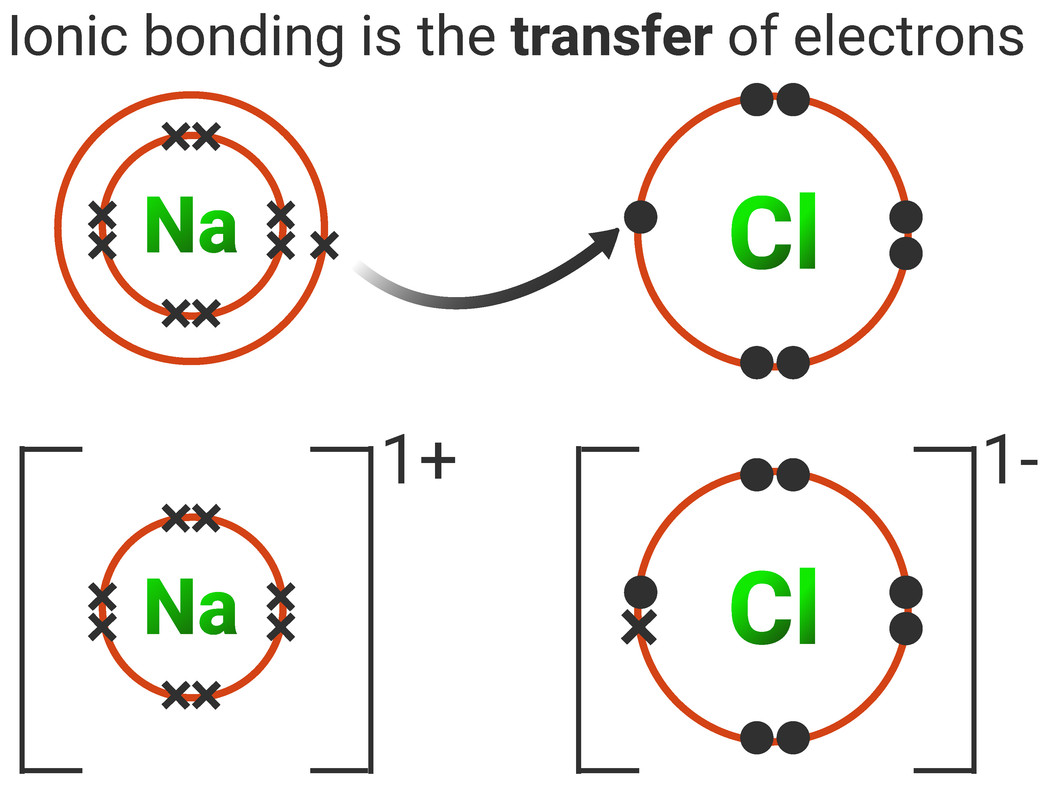
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk
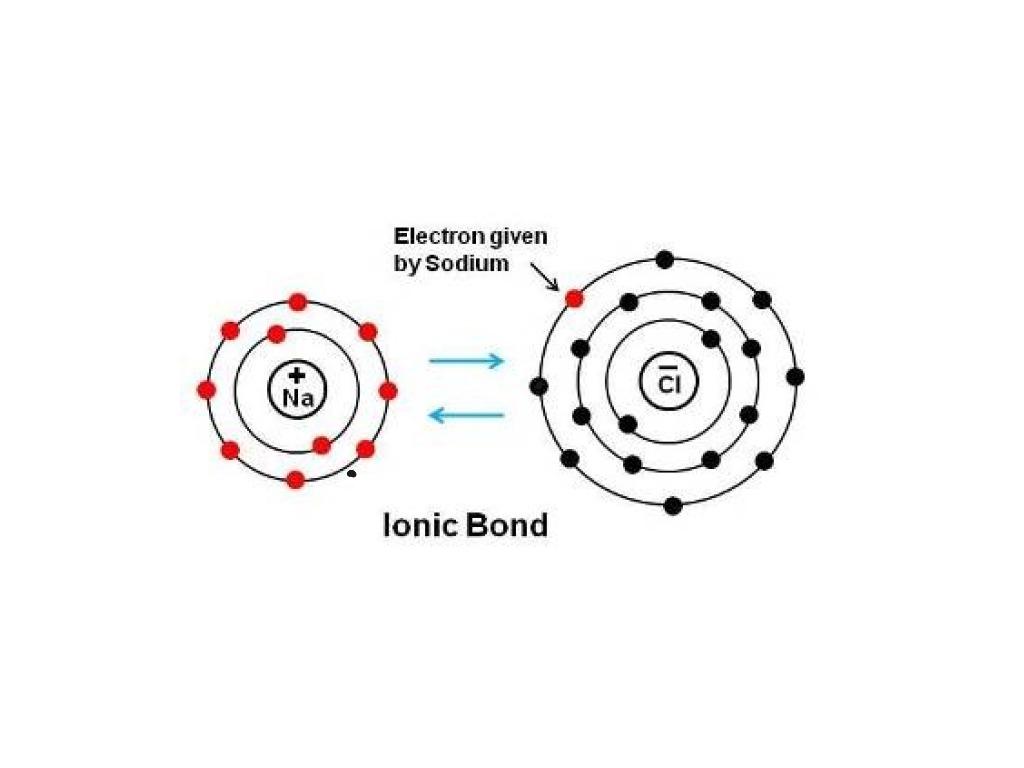
Ionic bond Science, Chemistry, Chemical Bonds ShowMe
Draw The Outer Shell Of Each Atom.
Lewis Diagram Of Formaldehyde (Ch₂O)
Draw The Electron Configuration Diagram For Each Atom.
In Section 4.7, We Demonstrated That Ions Are Formed By Losing Electrons To Make Cations, Or By Gaining Electrons To Form Anions.
Related Post:
.PNG)