Drawing Covalent Compounds
Drawing Covalent Compounds - How do you draw the lewis structure for ionic compounds? Complete the octets for any group 17 elements with lone pairs. Web two easy methods to draw structures of covalent molecules and assign geometries for each structure. The compound is often added to otherwise. For covalent bonding, we often want to draw how the atoms share electrons in the molecule. Web draw lewis structures for covalent compounds. Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons in a covalent bond. The example is for the nitrate ion. A lewis structure is a diagram that shows the chemical bonds between atoms in a. The following procedure can be used to construct lewis electron structures for more complex molecules and. Complete the octets for any group 17 elements with lone pairs. The following procedure can be used to construct lewis electron structures for more complex molecules and. Web draw a skeleton with one bond between each pair of atoms which are covalently bonded. How do you draw the lewis structure for ionic compounds? The example is for the nitrate ion. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. What are some examples of lewis structures? For example, consider ccl4 and nf3 as. Drawing every bond and every atom is tedious,. They are somewhat but less helpful for describing ions. Step by step examples for each method. How do you draw the lewis structure for ionic compounds? For covalent bonding, we often want to draw how the atoms share electrons in the molecule. The example is for the nitrate ion. The compound is often added to otherwise. The following procedure can be used to construct lewis electron structures for more complex molecules and. For covalent bonding, we often want to draw how the atoms share electrons in the molecule. Lewis structures are representations of molecules that include not only what atoms are present in the molecule but also how the atoms are connected. The example is for. Ethanethiol, c a 2 h a 6 s , is a clear liquid with a strong odor. Web here are the steps to draw a lewis structure. Web draw lewis structures for covalent compounds. Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons in a covalent bond. For covalent bonding, we. Draw lewis structures for covalent compounds. They are somewhat but less helpful for describing ions. Web draw lewis structures for covalent compounds. Web lewis dot structures are particularly useful for describing covalent bonding in compounds. Lewis structures are representations of molecules that include not only what atoms are present in the molecule but also how the atoms are connected. Web we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions. Web lewis dot structures are particularly useful for describing covalent bonding in compounds. Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons. Web in this video, we will go through how to draw lewis structures for covalent compound in five easy steps using carbon dioxide, co2, as an example. Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons in a covalent bond. Complete the octets for any group 17 elements with lone pairs.. What are some examples of lewis structures? Web draw a skeleton with one bond between each pair of atoms which are covalently bonded. How do you draw the lewis structure for ionic compounds? Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons in a covalent bond. Web here are the steps. Web in this video, we will go through how to draw lewis structures for covalent compound in five easy steps using carbon dioxide, co2, as an example. Web draw lewis structures for covalent compounds. How do you draw the lewis structure for ionic compounds? Web we also use lewis symbols to indicate the formation of covalent bonds, which are shown. Web here are the steps to draw a lewis structure. Web we also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions. Drawing every bond and every atom is tedious,. Step by step examples for each method. Web in the structures we’ve been drawing until now, a line between atoms has represented the two electrons in a covalent bond. The following procedure can be used to construct lewis electron structures for more complex molecules and. A lewis structure is a diagram that shows the chemical bonds between atoms in a. For covalent bonding, we often want to draw how the atoms share electrons in the molecule. How do you draw the lewis structure for ionic compounds? What are some examples of lewis structures? The compound is often added to otherwise. Ethanethiol, c a 2 h a 6 s , is a clear liquid with a strong odor. Web lewis dot structures are particularly useful for describing covalent bonding in compounds. Complete the octets for any group 17 elements with lone pairs. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. As a reminder, covalent bonding.
Covalent Compounds Examples and Properties
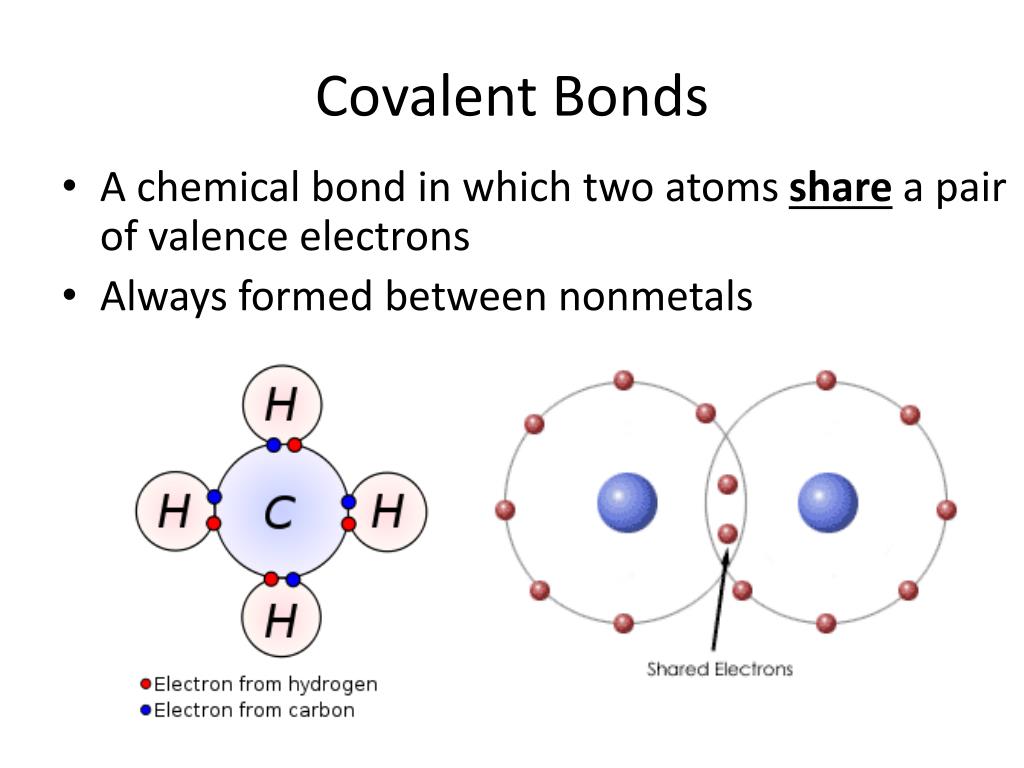
PPT Covalent Bonds PowerPoint Presentation, free download ID6647183
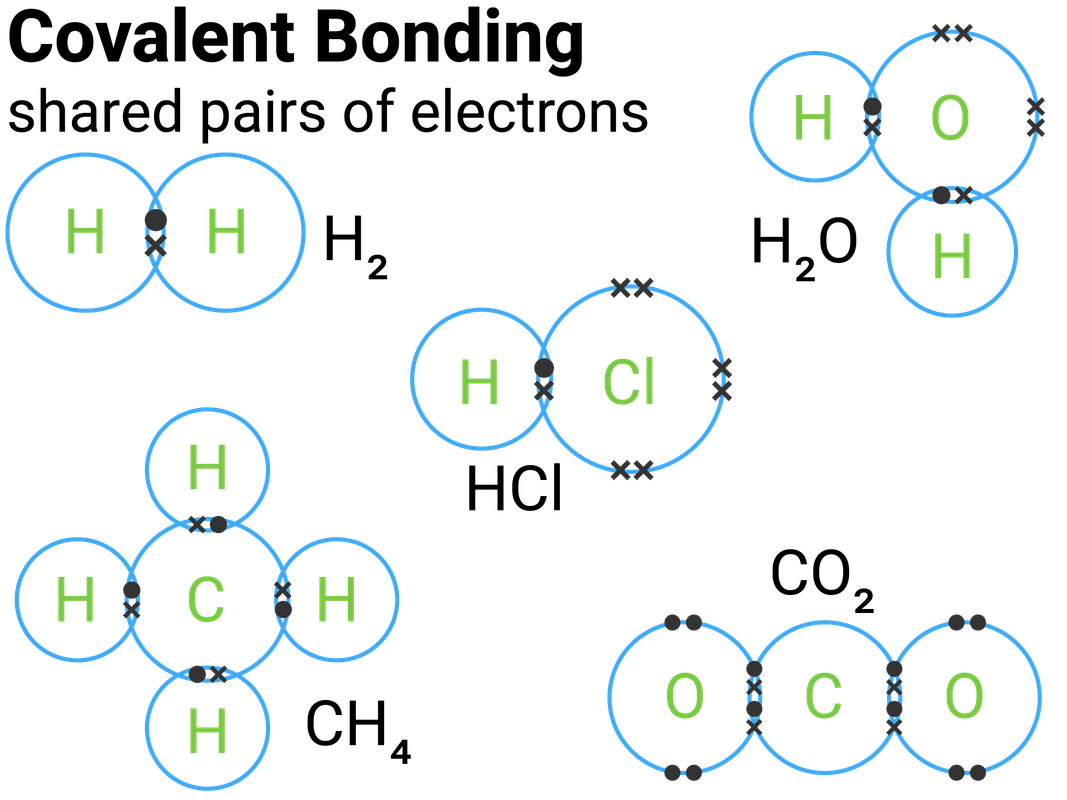
Bonding and Structure Edexcel T1 revisechemistry.uk

Drawing Covalent Bonds YouTube

How is a covalent bond formed

Lewis Diagrams of Covalent Compounds YouTube
Covalent Compounds

How to Draw Covalent Compounds YouTube

How to Draw Lewis Dot Structure of Covalent Compounds Chemical
/some-examples-of-covalent-compounds-603981_final21-a3faebbe543e404fb951d2e789031f56.jpg)
Examples of Covalent Bonds and Compounds
Web In This Video, We Will Go Through How To Draw Lewis Structures For Covalent Compound In Five Easy Steps Using Carbon Dioxide, Co2, As An Example.
Web Draw A Skeleton With One Bond Between Each Pair Of Atoms Which Are Covalently Bonded.
For Example, Consider Ccl4 And Nf3 As.
They Are Somewhat But Less Helpful For Describing Ions.
Related Post: