Draw The Lewis Structure Of Calcium Chloride
Draw The Lewis Structure Of Calcium Chloride - What kind of bond forms between a calcium atom and a chloride atom? Count all valence electrons available in cacl 2 in the first stage. The valence electrons are the electrons in the. The lewis structure was named after gilbert n. The astute reader may have noticed something: This problem has been solved! Identify the total number of valence electrons in the calcium atom and in each chlorine atom. This problem has been solved! Web this brief flash video shows how atoms of calcium and chlorine react to form the salt calcium chloride. A dash (or line) is sometimes used to indicate a shared pair of electrons: Web draw the lewis structure of calcium chloride. Calcium chloride (cacl2 or cl2ca) is an ionic compound. Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium: Become a study.com member to unlock this answer! Lewis, who introduced it in his 1916 article the atom and the molecule. This problem has been solved! Web cacl2 is a chemical formula for calcium chloride as it consists of one calcium and two chlorine atoms. Draw the lewis structure of calcium chloride. Calcium chloride (cacl2 or cl2ca) is an ionic compound. Select a tool to begin drawing. A dash (or line) is sometimes used to indicate a shared pair of electrons: There are 26 valence electrons. The astute reader may have noticed something: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The lewis structure was named after gilbert n. First, lets find the how many valence electrons chlorate has: Web this brief flash video shows how atoms of calcium and chlorine react to form the salt calcium chloride. Web the lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between. Calcium chloride (cacl2 or cl2ca) is an ionic compound. The structure of calcium chloride molecules is illustrated below. Web the lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). Web draw the lewis structure of calcium chloride. Select. A lewis structure is a way to show how atoms share electrons when they form a molecule. Web a lewis structure can be drawn for any covalently bonded molecule, as well as coordination compounds. 117k views 9 years ago. The valency of calcium is 2. First, lets find the how many valence electrons chlorate has: A dash (or line) is sometimes used to indicate a shared pair of electrons: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium: Draw the lewis structure of calcium chloride. Web the. Become a study.com member to unlock this answer! Web this brief flash video shows how atoms of calcium and chlorine react to form the salt calcium chloride. Lewis structures show all of the valence electrons in an atom or molecule. A lewis structure is a way to show how atoms share electrons when they form a molecule. X this problem. Chlorine belongs to the 17th group and the valence electron is seven. One calcium atom loses two electrons, so it. Lewis, who introduced it in his 1916 article the atom and the molecule. Is it polar, nonpolar, or ionic? Draw the lewis structure of calcium chloride. Find more chemistry widgets in wolfram|alpha. Web draw the lewis structure for calcium chloride? The valence electrons are the electrons in the. Web this brief flash video shows how atoms of calcium and chlorine react to form the salt calcium chloride. Chlorine belongs to the 17th group and the valence electron is seven. Lewis, who introduced it in his 1916 article the atom and the molecule. The valency of calcium is 2. Here’s the best way to solve it. Become a study.com member to unlock this answer! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In section 4.7, we demonstrated that ions are formed by losing electrons to make cations, or by gaining electrons to form anions. 117k views 9 years ago. Draw the lewis structure of calcium chloride. We are asked to draw the lewis structure for calcium chloride. A lewis structure is a way to show how atoms share electrons when they form a molecule. A single shared pair of electrons is called a single bond. This problem has been solved! Lewis symbols can also be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium: Identify the total number of valence electrons in the calcium atom and in each chlorine atom. Draw the lewis structure of calcium chloride. First, lets find the how many valence electrons chlorate has:
Draw the Lewis Structure of CaCl2 (calcium chloride) YouTube
![[DIAGRAM] Bohr Diagram For Calcium Chloride](https://i.ytimg.com/vi/9fWF5cQX9lg/maxresdefault.jpg)
[DIAGRAM] Bohr Diagram For Calcium Chloride
Calcium Chloride Lewis Dot Structure
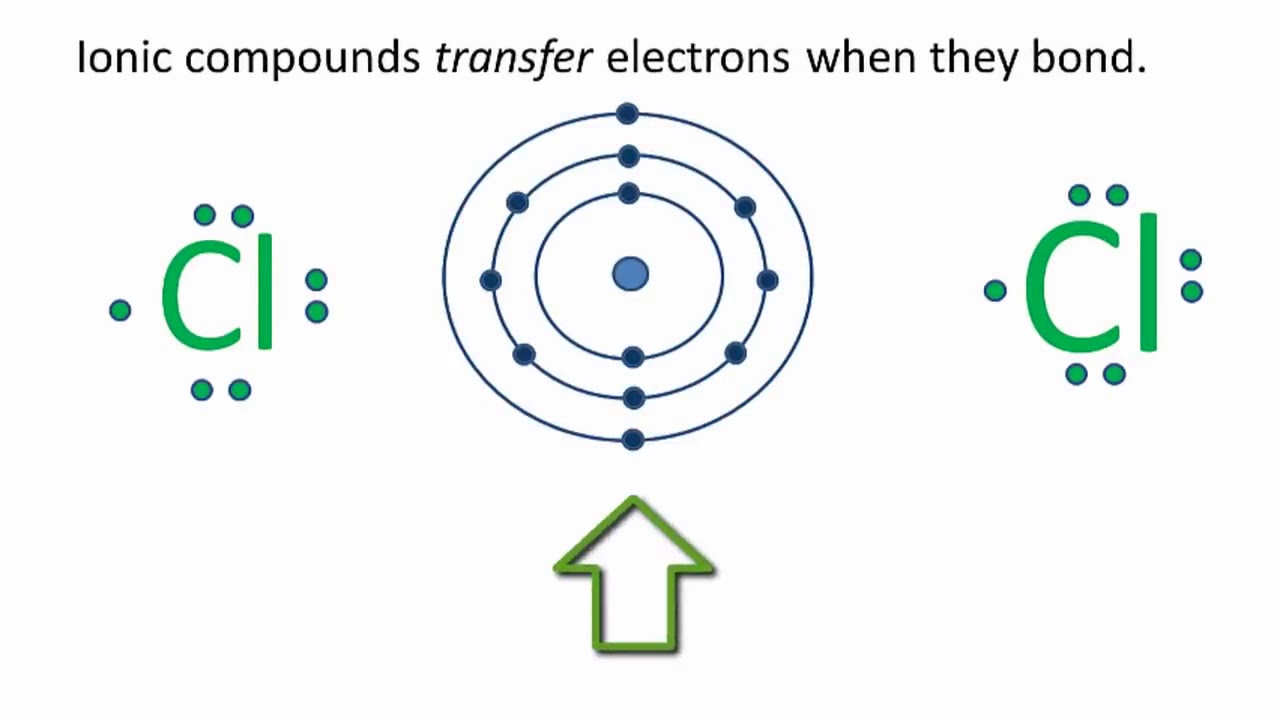
CaCl2 Lewis Structure How to draw the Lewis Dot Structure for Calcium

CaCl2 Lewis Structure (Calcium Chloride) YouTube

Electron Dot Diagram For Cacl2
Cacl2 Lewis Structure How To Draw The Lewis Dot Structure
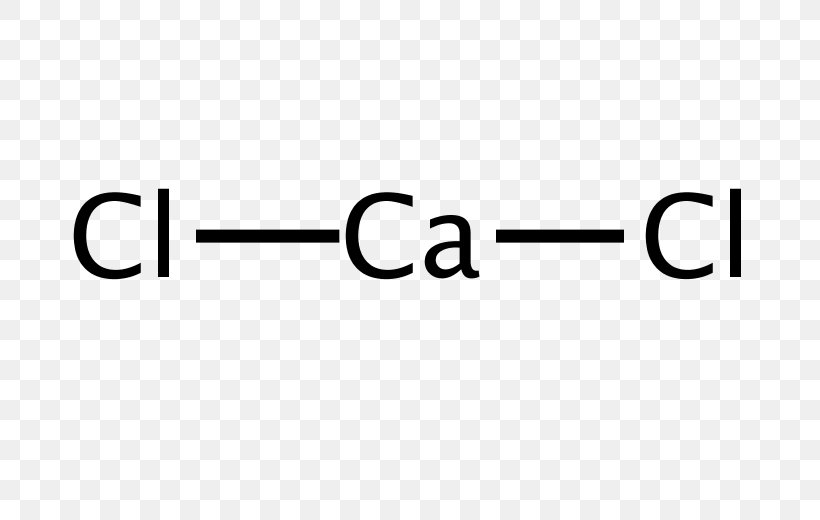
Calcium Chloride Lewis Structure Chemistry, PNG, 696x520px, Calcium

Calcium Chloride Lewis Structure
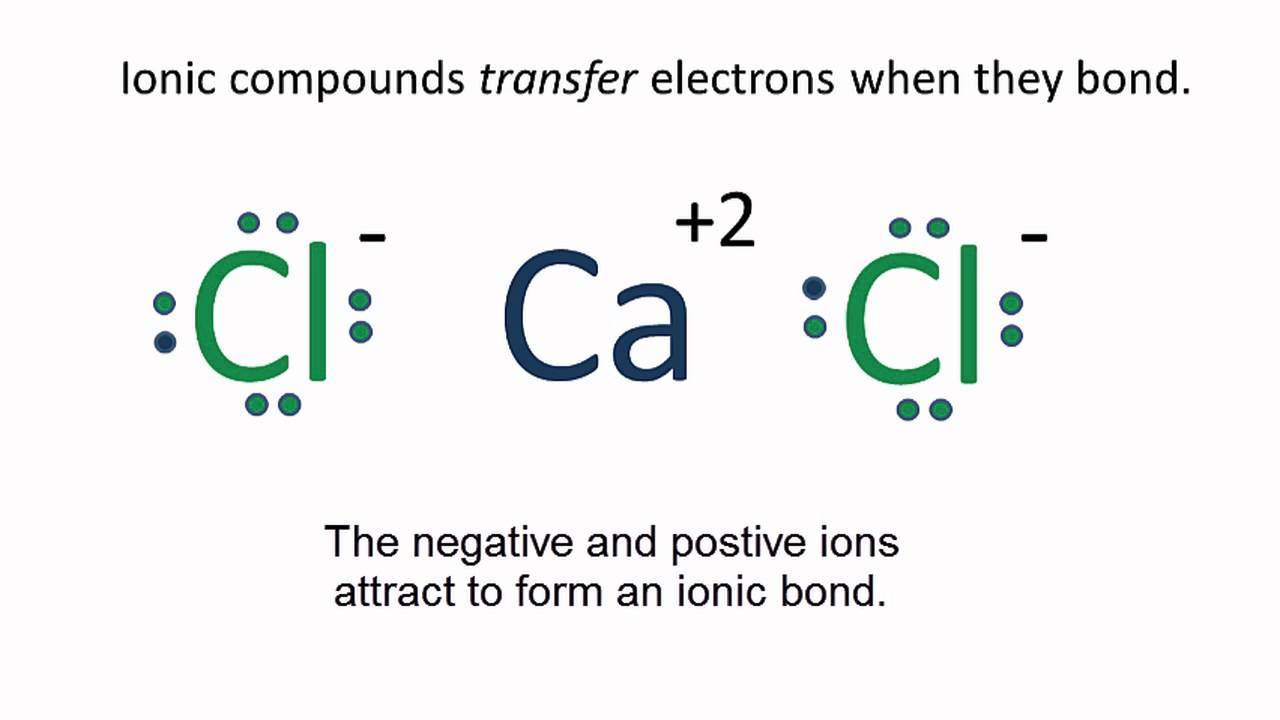
CaCl2 Lewis Structure How to draw the Lewis Dot Structure for Calcium
Count All Valence Electrons Available In Cacl 2 In The First Stage.
Calcium Chloride (Cacl2 Or Cl2Ca) Is An Ionic Compound.
Get The Free Lewis Structure Finder Widget For Your Website, Blog, Wordpress, Blogger, Or Igoogle.
Added Jun 9, 2014 By Webtester In Chemistry.
Related Post: