Draw The Lewis Structure For A Sulfur Monoxide So Molecule
Draw The Lewis Structure For A Sulfur Monoxide So Molecule - (valence electrons are the number of electrons present in the outermost shell of an atom). Submitted by robert v., oct. The so molecule has a triplet ground state similar to o 2 and s 2, that is, each molecule has two unpaired electrons. Send feedback | visit wolfram|alpha. Web therefore, a lewis structure must be drawn for a covalent molecule before its chemical formula can be determined. And to help you understand the lewis. #2 next, indicate lone pairs on the atoms. A each molecule of sulfur monochloride has two sulfur atoms and two chlorine atoms. S2o is a chemical formula for disulphur monoxide. This problem has been solved! Web added jun 9, 2014 by webtester in chemistry. To draw the lewis structure for the sulfur monoxide (so) molecule, start by drawing sulfur and oxygen bonded together. In order to draw the lewis structure of so, first of all you have to find the total number of valence electrons present in the so molecule. Web draw the lewis structure. Web therefore, a lewis structure must be drawn for a covalent molecule before its chemical formula can be determined. Draw lewis structures depicting the bonding in simple molecules. A each molecule of sulfur monochloride has two sulfur atoms and two chlorine atoms. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed earlier.. 495 views 10 months ago lewis structure. So, let’s calculate this first. In this case, we can condense the last few steps, since not. Try numerade free for 7 days. #1 draw a rough sketch of the structure. Web compute formal charges for atoms in any lewis structure; Draw the lewis structure for sulfur monoxide (so) molecule. The s−o bond length of 148.1 pm is. Thus far, we have discussed the. Web the molecular formula lists the elements in the molecule and the number of atoms of each. Thus far, we have discussed the. And to help you understand the lewis. Web draw lewis structures depicting the bonding in simple molecules. In order to draw the lewis structure of so, first of all you have to find the total number of valence electrons present in the so molecule. Web so lewis structure | image: Draw lewis structures depicting the bonding in simple molecules. Try numerade free for 7 days. Draw the lewis structure for sulfur monoxide (so) molecule. Web general chemistry 1. Web to properly draw the s 2 o lewis structure, follow these steps: 495 views 10 months ago lewis structure. The so molecule has a triplet ground state similar to o 2 and s 2, that is, each molecule has two unpaired electrons. Here, the given molecule is so (sulfur monoxide). Web compute formal charges for atoms in any lewis structure; Draw the lewis structure for sulfur monoxide (so) molecule. Try numerade free for 7 days. Use formal charges to identify the most reasonable lewis structure for a given molecule; Web to properly draw the s 2 o lewis structure, follow these steps: The so lewis structure represents the arrangement of a molecule consisting of one sulfur atom and one oxygen atom, also. Web draw the lewis structure for a. And to help you understand the lewis. Use formal charges to identify the most reasonable lewis structure for a given molecule; Submitted by robert v., oct. The so lewis structure represents the arrangement of a molecule consisting of one sulfur atom and one oxygen atom, also. Web we can draw the lewis structure of any covalent molecule by following the. The so molecule has a triplet ground state similar to o 2 and s 2, that is, each molecule has two unpaired electrons. We also use lewis symbols to indicate the formation of covalent bonds, which are shown in. Thus far, we have discussed the. S2o is a chemical formula for disulphur monoxide. (valence electrons are the number of electrons. Submitted by robert v., oct. Thus far, we have discussed the. 495 views 10 months ago lewis structure. Web to properly draw the s 2 o lewis structure, follow these steps: For example, consider fluorine and sulfur. You'll get a detailed solution from a subject matter expert that helps you learn. This problem has been solved! Web draw lewis structures depicting the bonding in simple molecules. S2o is a chemical formula for disulphur monoxide. To draw the lewis structure for the sulfur monoxide (so) molecule, start by drawing sulfur and oxygen bonded together. 12k views 2 years ago. Here, the given molecule is so (sulfur monoxide). The so lewis structure represents the arrangement of a molecule consisting of one sulfur atom and one oxygen atom, also. Try numerade free for 7 days. Send feedback | visit wolfram|alpha. Draw lewis structures depicting the bonding in simple molecules.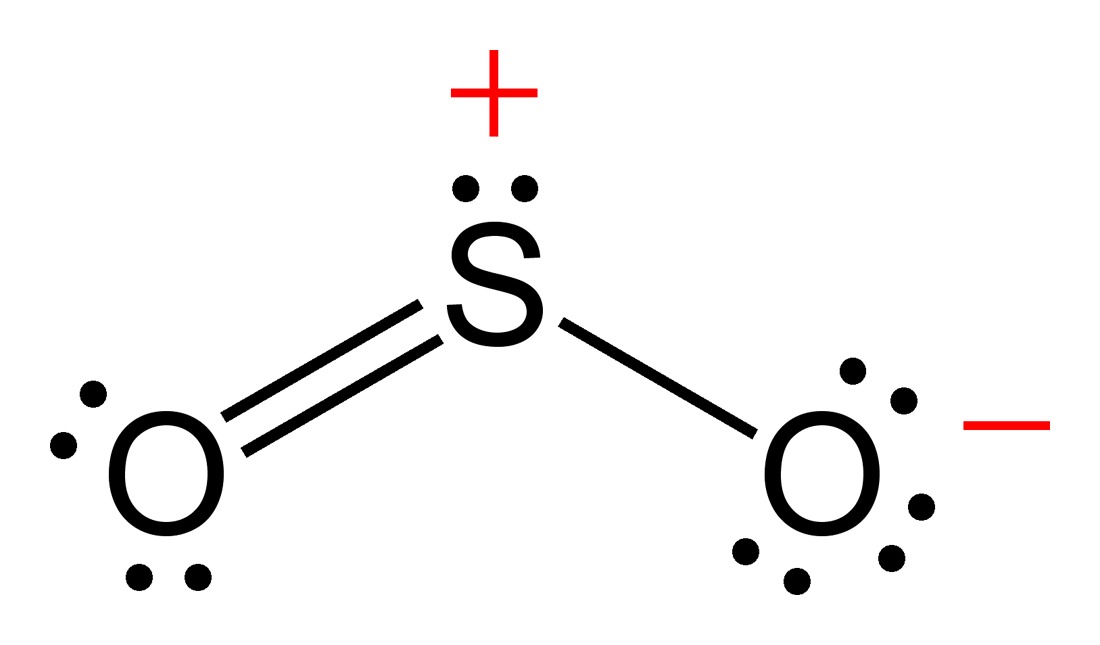
SO2(Sulfur Dioxide) Molecular Geometry & Lewis Structure Geometry of

SOLVED Draw the Lewis structure for sulfur monoxide (SO) molecule
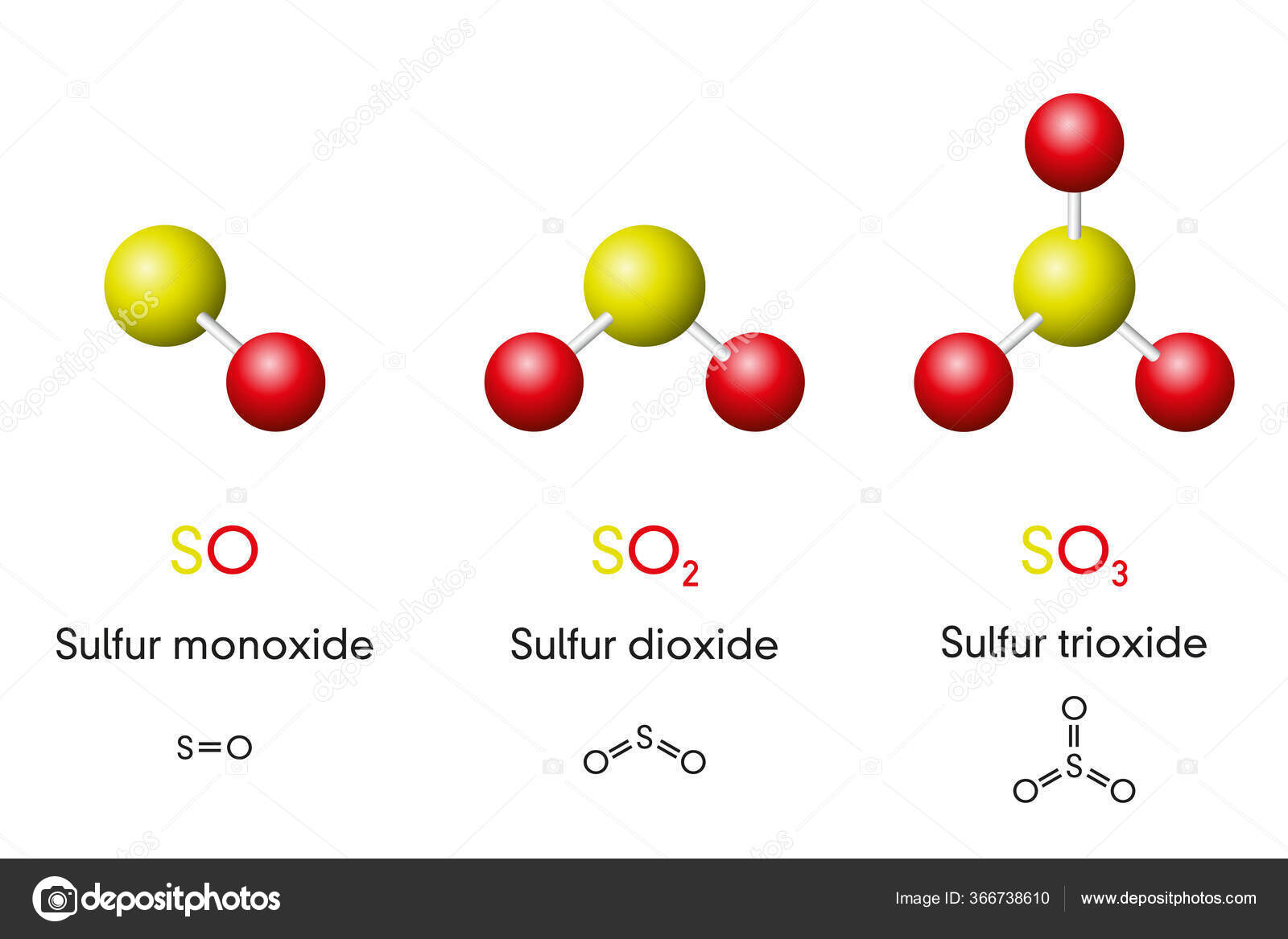
Three Sulfur Oxides Molecule Models Chemical Formulas Sulfur Monoxide

How to Write the Formula for Sulfur monoxide YouTube
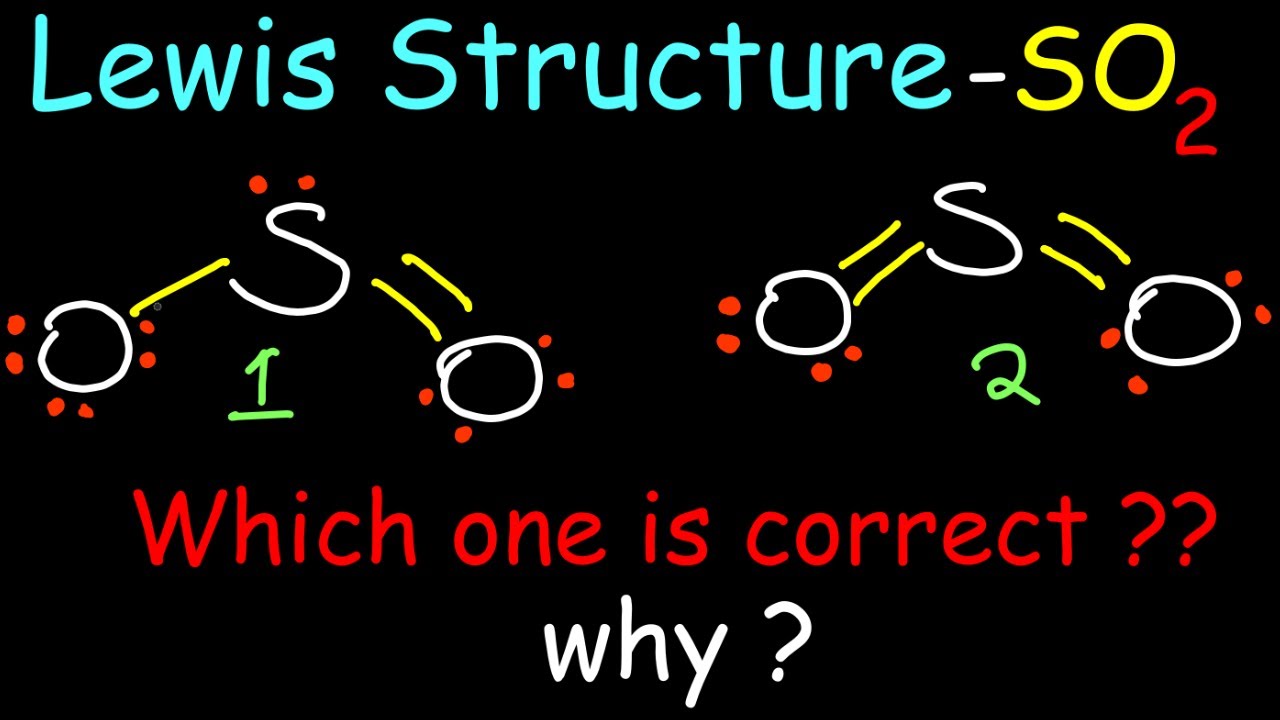
Lewis Structure of Sulphur Dioxide SO2 YouTube
Lewis Dot Diagram For Sulfur Wiring Diagram

So sulfur monoxide molecule Royalty Free Vector Image

Sulfur monoxide YouTube

Draw the Lewis Structure for a Sulfur Monoxide So Molecule

Draw the Lewis Structure for a Sulfur Monoxide So Molecule
Write Lewis Symbols For Neutral Atoms And Ions.
In Order To Draw The Lewis Structure Of So, First Of All You Have To Find The Total Number Of Valence Electrons Present In The So Molecule.
(Valence Electrons Are The Number Of Electrons Present In The Outermost Shell Of An Atom).
Chemical Bonding And Molecular Geometry.
Related Post:
