Draw All Resonance Structures For The Nitrate Ion No3
Draw All Resonance Structures For The Nitrate Ion No3 - In resonance structures, it does not require to show transformation of electrons by arrows. Draw a nitrogen atom (n) in the center surrounded by three oxygen (o) atoms: Select draw rings more erase n o. Web resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Here’s the best way to solve it. Organic chemistry resonance what is resonance? 98% (43 ratings) share share. Include electron lone pairs, and any formal charges. The nitrate ion has three resonance contributors. You can do this by (1) drawing the electron configuration per element or (2) consulting your. Draw all resonance structures for the nitrate ion, no3 • explicitly draw all h atoms. Web the carbonate (\(co_3^{2−} \)) ion; Select draw rings more erase n o. It compares and contrasts two or more possible lewis structures that can represent a particular molecule. Web drawing correct lewis structure is important to draw resonance structures. Organic chemistry resonance what is resonance? 152k views 3 years ago. Select draw rings more erase n o. 2k views 1 year ago. • include all valence lone pairs in your answer. In another tutorial, we learn how to draw resonance structures of nitrate ion. Web resonance structures are used when a single lewis structure cannot fully describe the bonding; In following examples, arrows are used to show electrons transformation. Organic chemistry resonance finding resonance structures. Draw a nitrogen atom (n) in the center surrounded by three oxygen (o) atoms: It compares and contrasts two or more possible lewis structures that can represent a particular molecule. You can decide this from looking about whether atoms have lone pairs or double bonds. Total number of electrons of the valance shells of nitrogen and oxygen atoms and charge of the anion. Select draw rings more erase n o. How to draw lewis. 20k views 2 years ago. 2k views 1 year ago. Web chemistry questions and answers. • include all valence lone pairs in your answer. Web the carbonate (\(co_3^{2−} \)) ion; Calculate the formal charge on all four atoms on one of thestructures. Draw the lewis structures for three resonance forms of the nitrate ion, no3. You can do this by (1) drawing the electron configuration per element or (2) consulting your. Draw a nitrogen atom (n) in the center surrounded by three oxygen (o) atoms: The concept was introduced in. First is you need to know the number of valence electrons. Here is how you can draw them: The concept was introduced in 1899 by johannes thiele in his “partial valence hypothesis” to explain benzene’s exceptional stability, which contradicted august kekulé’s 1865 structure with alternating single and double bonds. 2k views 1 year ago. Web chemistry questions and answers. Here is how you can draw them: We draw lewis structures to predict: You can decide this from looking about whether atoms have lone pairs or double bonds. Web to draw all resonance structures, take the lewis structure we drawn by using vespr rule. Total number of electrons of the valance shells of nitrogen and oxygen atoms and charge of. We draw lewis structures to predict: Here’s the best way to solve it. • do not draw double bonds to oxygen unless they are needed for the central atom to obey the. By exchanging the electron pairs around the oxygens. This problem has been solved! In another tutorial, we learn how to draw resonance structures of nitrate ion. • include all valence lone pairs in your answer. We draw lewis structures to predict: 2k views 1 year ago. By exchanging the electron pairs around the oxygens. There are one nitrogen atom and three oxygen atoms in the nitrate ion. How to draw lewis structures: Draw a nitrogen atom (n) in the center surrounded by three oxygen (o) atoms: Here is how you can draw them: In following examples, arrows are used to show electrons transformation. Web resonance structures are used when a single lewis structure cannot fully describe the bonding; In another tutorial, we learn how to draw resonance structures of nitrate ion. Draw all resonance structures for the nitrate ion, no3 • explicitly draw all h atoms. 2k views 1 year ago. 152k views 3 years ago. Organic chemistry resonance what is resonance? The concept was introduced in 1899 by johannes thiele in his “partial valence hypothesis” to explain benzene’s exceptional stability, which contradicted august kekulé’s 1865 structure with alternating single and double bonds. In resonance structures, it does not require to show transformation of electrons by arrows. Draw the lewis structures for three resonance forms of the nitrate ion, no3. • do not include overall ion charges or formal charges in your drawing. Web each of stable resonance structure of nitrate ion is a lewis structure.
Lewis structure of NO3 (Nitrate ion)Draw the Lewis dot structure of
[Solved] 2. Draw four resonance forms of the nitrate ion NO3

Resonance Structures of NO3(1), nitrate ion YouTube
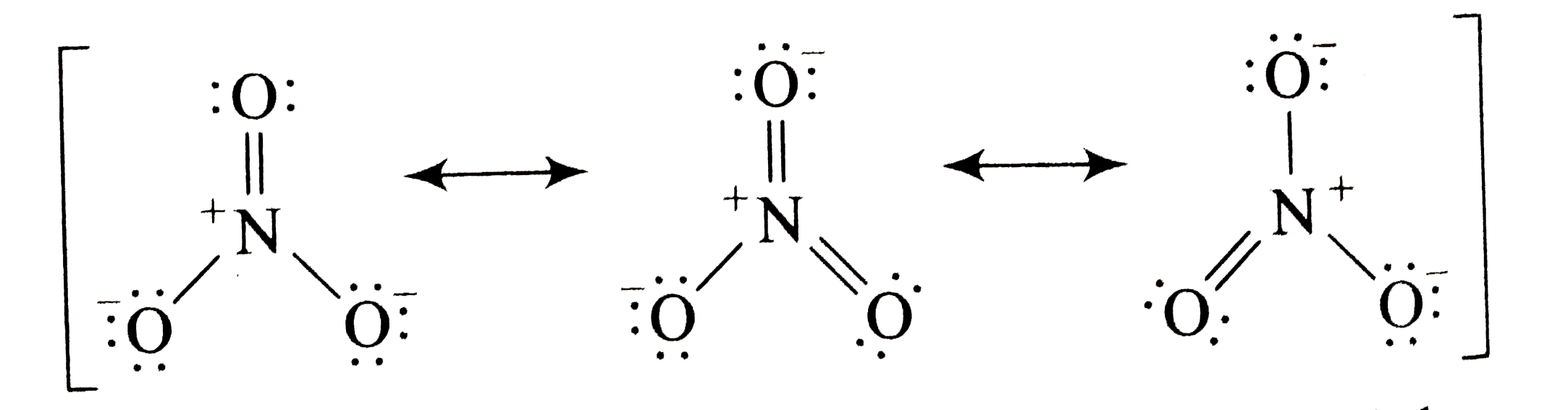
Nitrate Ion Lewis Structure No3
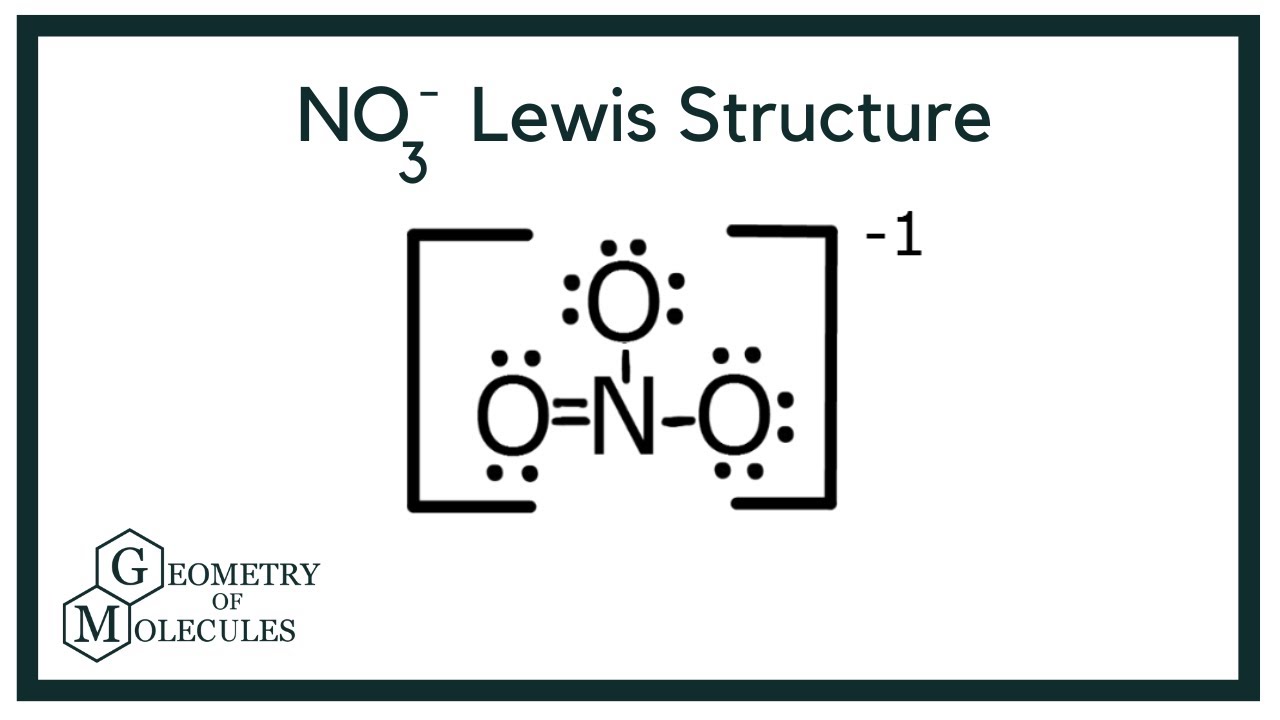
NO3 Lewis Structure Draw Lewis Dot Structure of Nitrate Ion YouTube
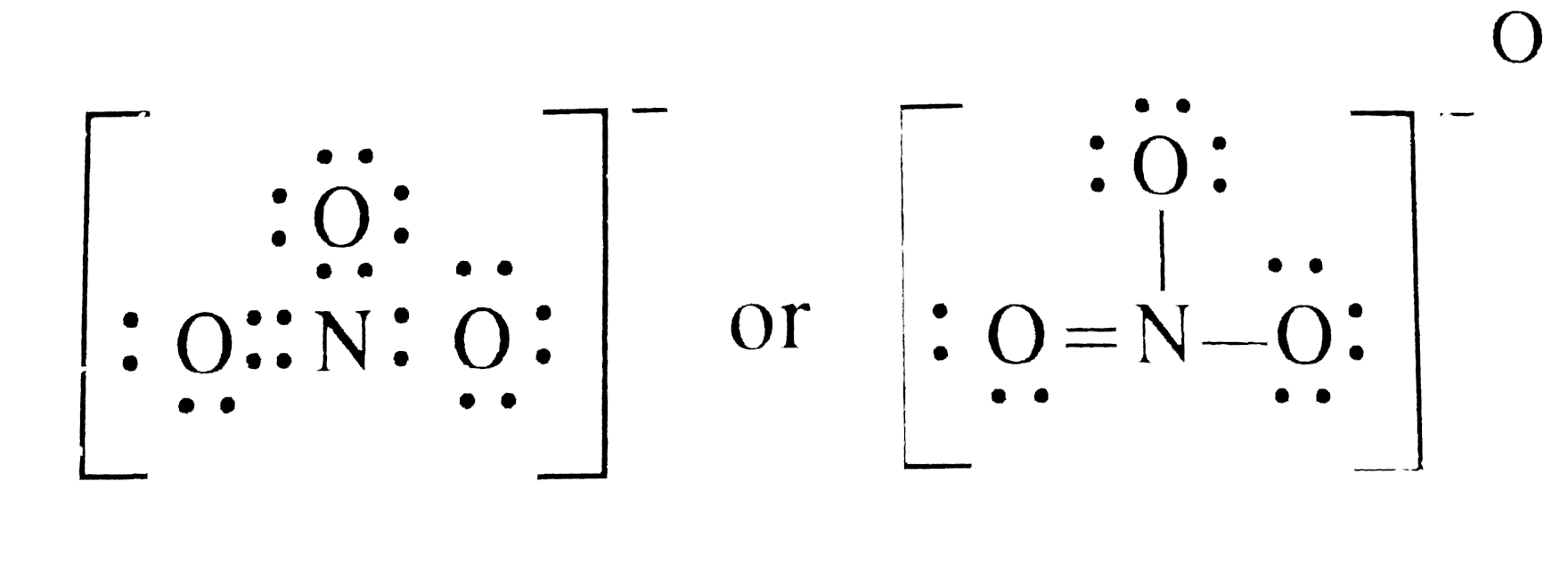
How To Draw The Lewis Dot Structure For No3 Nitrate Ion
[Solved] Draw all valid Lewis structures for the nitrate ion, showing

what is resonance?resonating structure of NO3 ion Brainly.in
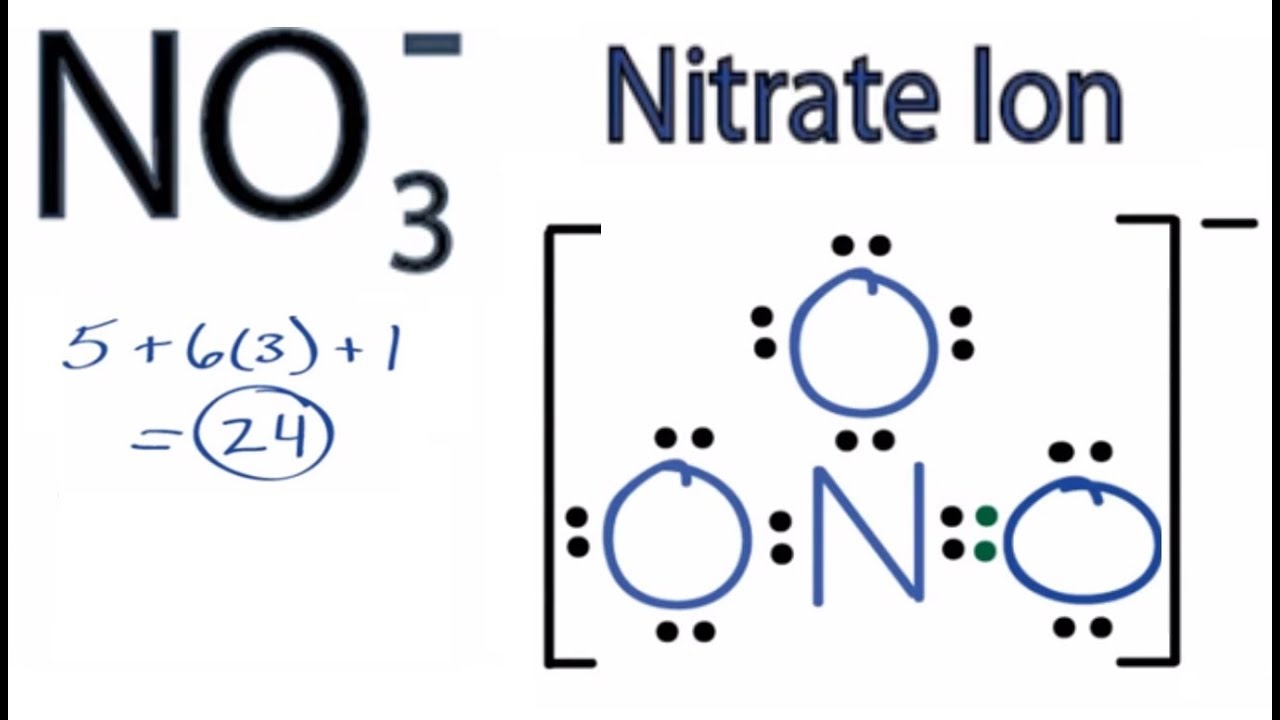
Nitrate Ion Lewis Structure How to Draw the Lewis Structure for
[Solved] Draw Lewis structure(s) for the nitrate ion ( NO 3 ). If
Determine The Total Number Of Valence Electrons In The Molecule.
Therefore, We Can Draw Resonance Structures.
• Do Not Include Overall Ion Charges Or Formal Charges In Your Drawing.
Calculate The Formal Charge On All Four Atoms On One Of Thestructures.
Related Post: