Boiling Point Drawing
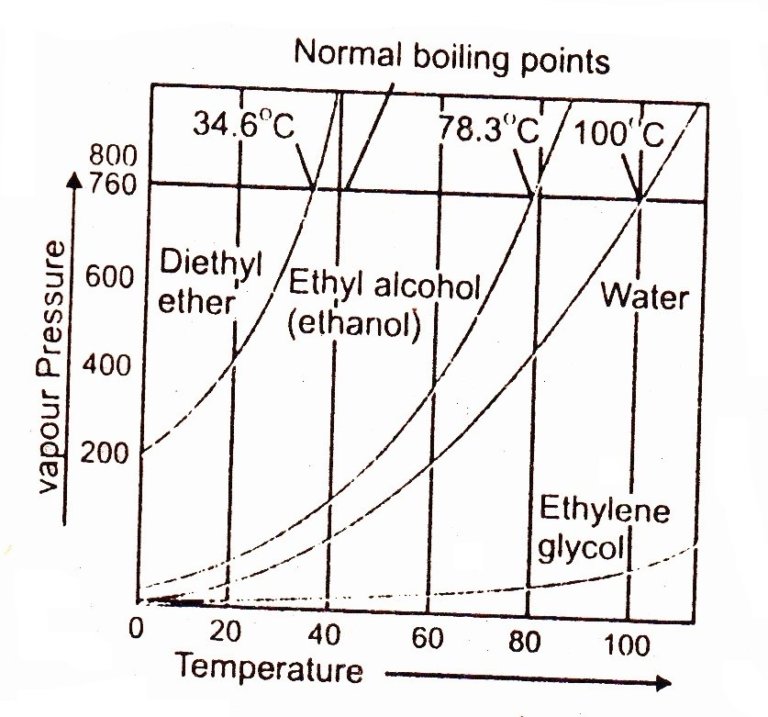
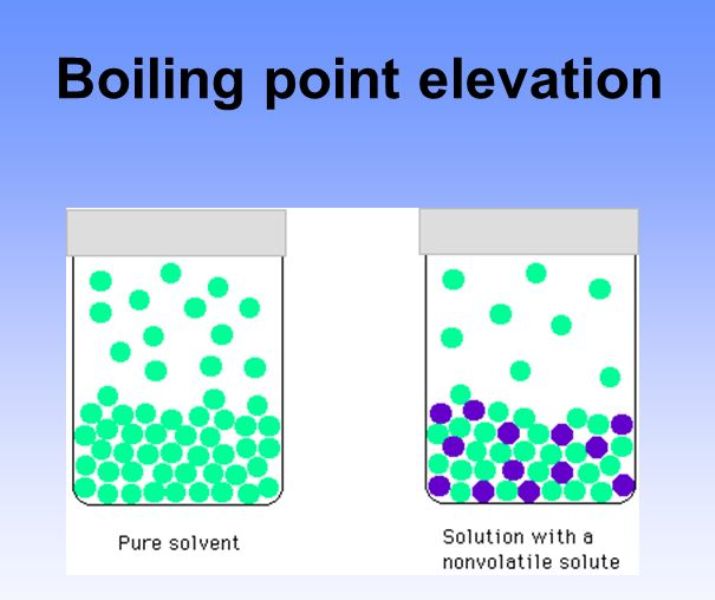
Boiling Point Drawing - The boiling point is the temperature at which boiling occurs for a specific liquid. Web boiling points are a measure of intermolecular forces. 2.9k views 1 year ago. Web the boiling points of various liquids can be illustrated in a vapor pressure curve (figure below). T (of) = [t (oc)] (9/5) + 32. As solute molecules are added to water, the boiling point increases. Under this condition, addition of heat results in the transformation of the liquid into its vapor without raising the temperature. Thunderstorms have rising and sinking currents of air called. The overarching principle involved is simple: The most straightforward method uses a thiele tube, and has the advantage of using less than 0.5ml 0.5 ml of material. Boiling point and melting point in organic chemistry. The overarching principle involved is simple: The strength of intermolecular forces (and therefore impact on boiling points) is ionic > hydrogen bonding > dipole dipole > dispersion; A vapor pressure curve is a graph of vapor pressure as a function of temperature. The boiling point is the temperature at which boiling occurs. Web determining boiling points from structure. How strong are intermolecular forces within a pure sample of the substance? Web the formal definition in science is that boiling point is the temperature where the vapor pressure of a liquid equals the vapor pressure of its environment. You could heat each of the liquids and measure their boiling points with a thermometer.. Explain why you think this constitutional isomer would have a higher boiling point. Order these compounds from lowest to highest boiling point: The boiling point of a liquid depends on temperature, atmospheric pressure, and the vapor pressure of the. Chemical makeup = structure and bond type. Web the boiling point is defined as the temperature at which the saturated vapor. Web the boiling points of various liquids can be illustrated in a vapor pressure curve (figure below). Hexane has six carbons, one, two, three, four, five, and six. How strong are intermolecular forces within a pure sample of the substance? Can you determine the concentration of solute molecules in each of the three liquids? Therefore, we can compare the relative. Web the formal definition in science is that boiling point is the temperature where the vapor pressure of a liquid equals the vapor pressure of its environment. The most straightforward method uses a thiele tube, and has the advantage of using less than 0.5ml 0.5 ml of material. The boiling point of a liquid depends on temperature, atmospheric pressure, and. For general purposes it is useful to consider temperature to be a measure of the kinetic energy of all the atoms and molecules in a given system. Web intermolecular forces (imfs) can be used to predict relative boiling points. What is boiling point elevation? As solute molecules are added to water, the boiling point increases. Web boiling points are a. Let's draw in another molecule of pentane right here. The strength of intermolecular forces (and therefore impact on boiling points) is ionic > hydrogen bonding > dipole dipole > dispersion; The boiling point is the temperature at which boiling occurs for a specific liquid. Web draw a constitutional isomer of this molecule that would be likely to have a higher. Web determining boiling points from structure. How strong are intermolecular forces within a pure sample of the substance? Boiling point elevation refers to the increase in the boiling point of a solvent upon the addition of a solute. Temperature given as °c, °f, k and °r. And pentane has a boiling point of 36 degrees celsius. The most straightforward method uses a thiele tube, and has the advantage of using less than 0.5ml 0.5 ml of material. A vapor pressure curve is a graph of vapor pressure as a function of temperature. Can you determine the concentration of solute molecules in each of the three liquids? What is boiling point elevation? What is boiling point diagram? The boiling point at atmospheric pressure (14.7 psia, 1 bar absolute) for some common fluids and gases can be found from the table below: Let's draw in another molecule of pentane right here. Web pentane has five carbons, one, two, three, four, five, so five carbons for pentane. Web phase diagram is a graphical representation of the physical states of. As we cross the lines or curves on the phase diagram, a phase change occurs. Boiling point elevation refers to the increase in the boiling point of a solvent upon the addition of a solute. Draw a different conformation of this constitutional isomer. And pentane has a boiling point of 36 degrees celsius. Can you determine the concentration of solute molecules in each of the three liquids? For example, for water, the boiling point is 100ºc at a pressure of 1 atm. T (of) = [t (oc)] (9/5) + 32. Web the boiling point is defined as the temperature at which the saturated vapor pressure of a liquid is equal to the surrounding atmospheric pressure. The intermolecular forces increase with increasing polarization of bonds. Under this condition, addition of heat results in the transformation of the liquid into its vapor without raising the temperature. Web the boiling points of various liquids can be illustrated in a vapor pressure curve (figure below). Web arts by maryam arshad. A vapor pressure curve is a graph of vapor pressure as a function of temperature. With our boiling point calculator, you can quickly determine the atmospheric boiling point of various substances. At this temperature, the liquid changes into the vapor (gas) phase. The boiling point is the temperature at which boiling occurs for a specific liquid.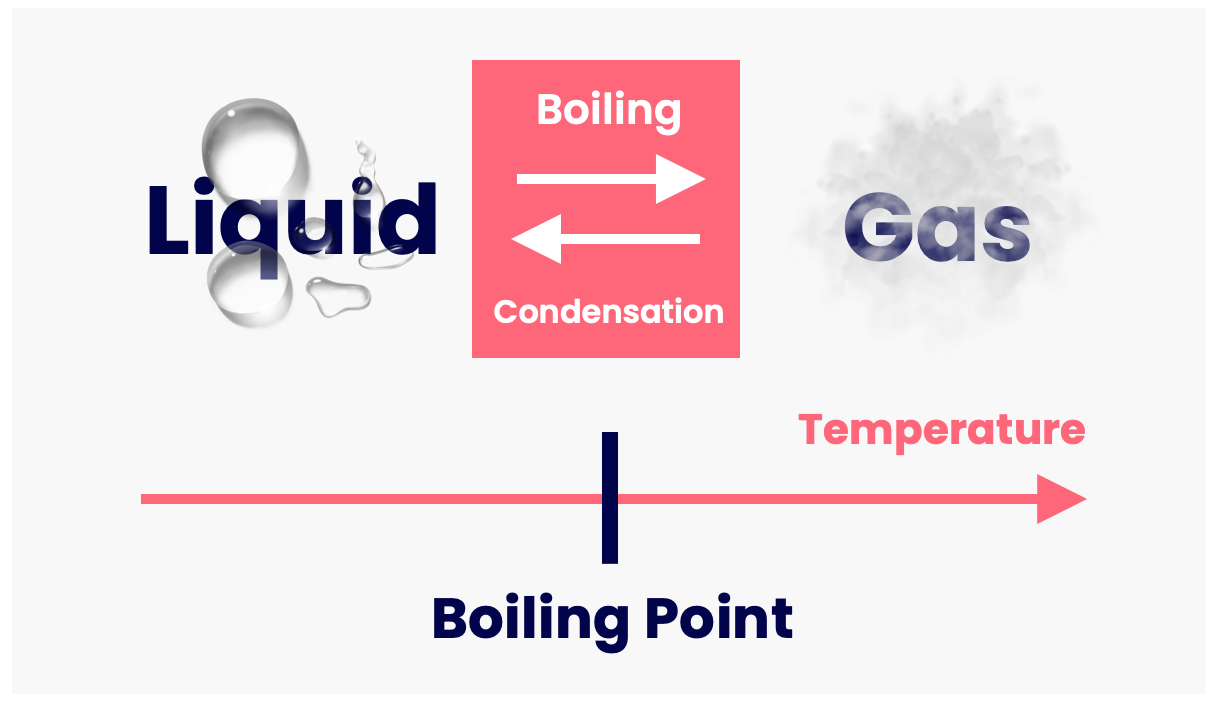
Melting & Boiling • Matter • Physics Fox
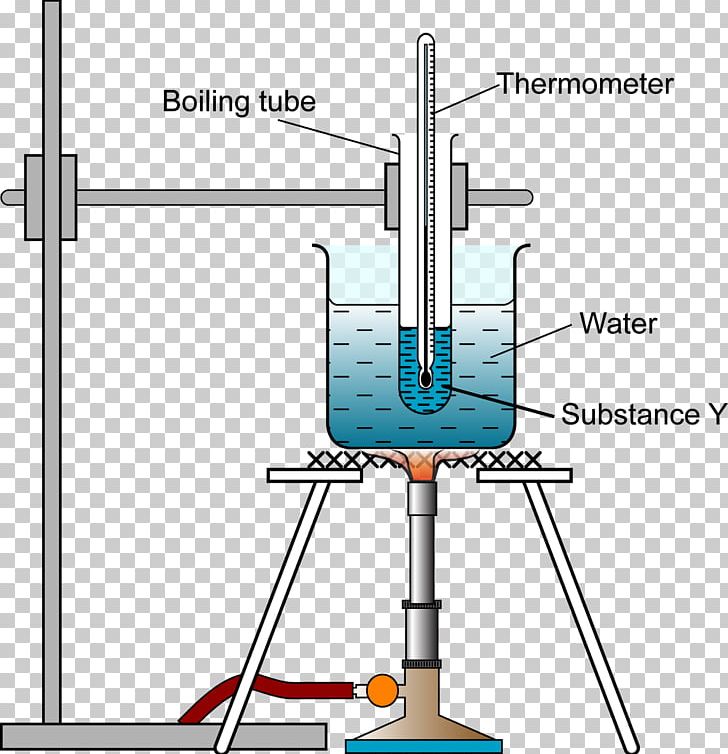
Boiling Point Melting Point Heat Temperature Chemistry PNG, Clipart

Boiling Point of Water What Temperature Does Water Boil?

Boiling point Melting point Heat Temperature Chemistry, laboratory
Definition and Explanation of Boiling Point Chemistry Skills

Vapour pressure, boiling point, properties of liquids Chemistry

Boiling Point Definition of Boiling Point

Boiling Point and Melting Point in Organic Chemistry Chemistry Steps

Thermal Secrets to Boiling Point Calibration
Boiling Point Examples in Everyday Life StudiousGuy
You Could Heat Each Of The Liquids And Measure Their Boiling Points With A Thermometer.
Web Determining Boiling Points From Structure.
Boiling Point And Melting Point In Organic Chemistry.
The Boiling Point Of A Liquid Varies Depending Upon The Surrounding Environmental Pressure.
Related Post: